Pooling of asymptomatic SARS COV-2 COVID-19 samples for (PCR/or other) testing
Contents
- Introduction
- Testing protocol section
- Procedural risk assessment: to be completed by laboratory according to in-house process
- Other safety information
Appendix: Publications and further literature
Classification: Official
Publications approval reference: C0777
Standard operating procedure
Pooling of asymptomatic SARS COV-2 COVID-19 samples for (PCR/or other) testing
25 September Version 1
This guidance is correct at the time of publishing. However, as it is subject to updates, please use the hyperlinks to confirm the information you are disseminating to the public is accurate.
1. Introduction
Purpose and scope
The purpose of this document is to describe the laboratory procedure for conventional pooling.
Rationale
To support the urgent increase of rapid SARS-COV-2 (COVID-19) PCR (or other technology) testing, to ensure the time to turn samples around does not exceed 24h (which translates into problems with patient flow, and potential unnecessary patient and staff exposure), and to ensure the most effective and efficient use of reagents and equipment, there is a need for further consideration of alternative testing processes to be deployed. It is also expected that as winter approaches the demand for COVID-19 testing will increase, putting more pressure on an already stretched system. Sample pooling strategies increase testing capacity when resources are limited. There is growing evidence to support this strategy (see Appendix 1) in the context of COVID-19.
What is sample pooling?
Sample pooling allows more people to be tested quickly using fewer testing resources. It does this by allowing samples from several people to be analysed in one test.
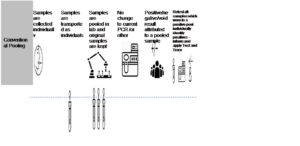
Sample pooling process:
- Collect individual samples (swabs or saliva according to test method validated) from a defined group of people, which may vary in size (usually between 6 to 12 people and is dependent on prevalence within the population)
- Samples are then mixed together (under biosafety containment) and become a ‘pooled” sample’ – this standard operating procedure (SOP) describes how this is to be done in the laboratory.
- All samples are then stored at 40c from individuals to enable retrieval, if retest is needed
- QPCR test (or other validated test) is performed on “pooled” samples
- If pooled sample is positive all individual samples in that “pool” group are tested individually
- If the pooled sample is negative assume all samples within the pool are negative
There is one single objective for pooling in the context of the UK National Testing Programme: to significantly increase testing capacity during a time of low background prevalence, reducing reagent consumption per test and increasing overall total testing capacity in the country.
The primary methodology to be used is conventional pooling – this can be applied to many populations where there is need and benefit from identifying positive individuals e.g. Asymptomatic patients and professionals, at a time of low positive prevalence. In most current publications pooling has been undertaken at a time where the positive rate in the population being tested is 8% positive with an estimated community positive rate in the range of 1% to 2%. If the rate of positive tests in the community rises, the use of pooling may need to be limited to use cases of lower positive prevalence or the pool size reduced. Mathematical modelling to calculate the optimal pool size given the prevalence or % positive samples in a testing population is well documented.1,2
Note that there are other approaches to pooling including cohort pooling, where samples are collected in small cohorts, pooled at source and tested. This is a strategy that may be used for Care Homes, or orthogonal pooling, which allows mathematical modelling to be applied to ascertain optimal pool sizes with samples appearing in more than one pool. These are not described in this SOP.
The future National Testing Strategy will be informed by both this pilot study and the current asymptomatic study being undertaken through the Testing Validation Group (TVG).
Responsibility
The head of laboratory is responsible for the implementation, validation, maintenance and review of this procedure. Individual members of staff are responsible for ensuring that they are familiar with and adhere to the procedures described.
It is the responsibility of all personnel trained to handle and test clinical samples suspected of containing SARS-COV-2 to be familiar with and in compliance with the procedures outlined in this document and references listed in Appendix 1. The Quality Manager is responsible for auditing the effectiveness of the procedures.
Advantages
Sample pooling enables more efficient testing at a time of low background prevalence; more patients are tested with fewer reagents. In times of low positive prevalence, the majority of individuals will have a Negative result, which will allow patients and staff flows to proceed with no transmission risk, whilst those smaller numbers in positive pools await confirmatory tests.
Disadvantages
There is a small risk of false negative results due to dilutional effect (seen on very weak positives); this can be minimised by limiting the pool size to no more than 12 samples in a pool.1,2 The optimum number of the pool size for any given technology will need to be determined by in-house verification. A rapid rise in the background prevalence can quickly lead to a higher requirement for testing capacity to confirm positive pools. Positive pools can take longer to confirm impacting upon turnaround times. Monitoring of the community prevalence and positivity rate are necessary to know when to stop using this strategy. Also, Laboratory based monitoring will be needed, if significant numbers of positives are seen in those tested this must also flag a change in approach. This monitoring should already be in place in NHS and PHE Laboratories.
When and where to use it?
Sample pooling is only efficient when the expected positivity rate is low. No pooling strategy is effective when the positivity rates exceed 10%. Aggressive pooling can be used with low transmission; however the pool size needs to be reduced quickly when the positivity rate is above 1% e.g. for a positivity rate of around 3% the ideal pool size is 6 samples. Lab processes must be in place to successfully and reliably identify individual samples from pooled samples and retest with lower TAT and accuracy. Suggested target group: asymptomatic elective patients and asymptomatic staff where most samples are going to be negative 3,4,5. Pooling should not be used on individuals who are symptomatic and likely to test positive. Pooling should not be used for individuals where a rapid confirmation of COVID-19 status is required, such as for diagnostic purposes or for testing to cohort patients in healthcare settings.
References: Also see Appendix 1
{INSERT items}
Definitions
The primary methodology to be used is conventional pooling (Figure 1) this can be applied to many populations where there is need and benefit from identifying positive individuals, eg asymptomatic patients and professionals, at a time of low positive prevalence. [No other pooling method should be used without prior consultation with NHS England and NHS Improvement COVID-19 testing cell.]
Figure 1: Conventional pooling
Period of review
Period of initial review
As this is a pilot, the process will be reviewed in 1 month following initiation, to see what needs to be amended. All results will also need to be shared with the Testing (Technology) Validation Group (TVG) through an agreed template, which will be developed by TVG and provided to pooling sites for completion.
Then after every month unless any amendments to processes, which may affect the end result. Note – Changes in background prevalence may lead to a requirement to undertake earlier review.
2. Testing protocol section
All laboratories should undertake verification of their SARS COV-2 PCR/or other assay protocols when adding pooling, to understand the change in Ct values when pooling is applied (from published data between 2.6–3.8 Ct value variance) and adjust threshold reporting values accordingly.
Samples should be collected in the laboratories validated tubes with use of validated swabs where appropriate for the standard QPCR or other technology being used for pooling assay.
Testing procedures
Prepare a log of samples to be pooled
- Before beginning, prepare a log of what samples are to be pooled together and assign a unique identifier to the pool
- Pools can contain between 6 and 12 samples as agreed following the modelling of the capacity required
- Pooling to conserve tests is recommended when positive test rates are below 10%
Prepare for testing
All methods and guidelines for preparing the Laboratory’s standard SARS COV-2 PCR/or other protocol should be followed. Proper use of personal protective equipment (PPE), safe handling of samples and test tubes or cartridges, waste disposal and cleaning up of spillages should be followed as described by the laboratory’s Health and Safety procedures for the standard SARS-COV-2 assay testing.
Combine samples into a pool
To prevent cross-contamination between samples, only a single patient sample tube should be open at any given time. Also, pooling can only take place if there is sufficient sample for the pooling assay and a retest, in the event that the pooled samples test positive.
- Working in a prepared secluded work area, thoroughly mix each patient sample to be pooled by inverting the tube five times.
- Open the secondary tube to be used for preparing the pool
- Perform the following steps for each patient sample to be pooled:
-
- Open lid carefully to avoid touching droplets on the inner lid
- Place lid upright (inner side up) to avoid droplets falling on the work surface
- Transfer 300 μL of the test sample into the secondary tube with either a mechanical P1000 pipette or a single-use transfer pipette
- Rinse the tip/transfer pipette in disinfectant and leave in the waste container. Do not use a tip / transfer pipette more than once
- Cap and rack the patient sample tube and save for storage
- Samples should be stored at 4⁰C for no longer than 24 hours before retest. Following retest samples can be stored according to standard laboratory practice.
- Thoroughly wipe gloves with approved disinfectant before handling the next patient sample
- Repeat the above steps with each patient sample included in the pool
4. Once all samples have been pooled, tightly cap the secondary tube and invert it 10 times to mix thoroughly
5. Revert back to Laboratory’s standard SARS COV-2 testing protocol
a. Proceed to loading the pool into standard tube/plate or on the cartridge as Laboratory’s protocol dictates.
Running samples and interpreting results
- All methods for creating a test run, generating reports, and interpreting results should be performed according to the SARS-COV-2 assay SOP.
- There should be a process in place to identify each individual tested in the pool and the appropriate action taken.
- For negative results, every individual in the pool will have a negative result reported according to the laboratory’s standard reporting protocol.
- For positive results, every individual in that pool will need to be urgently retested on the next run as a single assay according to laboratory’s standard operating protocol for SARS COV-2 assay, ensuring fastest TAT for those samples.
- All results to be logged into the LIMS system according to Laboratory standard practice
- All positive results to be notified to PHE though SGSS.
- There should be a process to monitor and note whether background prevalence is within defined bounds for the validated approach.
- Results to be reported back to TVG on template provided on a weekly basis.
3. Procedural risk assessment: to be completed by laboratory according to in-house process
Organisation and department name:
Procedure name:
Assessor: XXX Date: XXX To be reviewed: XXX
All risks to be considered including transport media, storage requirements of pathogens and process risks, and risk to breaching TAT, etc.
| Hazard | People at risk | Controls in place | Likelihood | Severity | Risk score | Controls adequate YES/NO | Action | Revised score | Lead responsibility | £’s | Completion date |
4. Other safety information
To be completed by laboratory: please include any other laboratory procedure safety information needed.
A separate copy of this risk assessment should be kept for reference with other risk assessments, Health and Safety files, and COSHH regulations, according to standard laboratory practice.
Fire Points
The nearest fire points for a CO2 fire extinguisher are: [To be completed by laboratory]
COSHH
[To be completed by laboratory]
Appendix: Publications and further literature
- Mallapaty S. The mathematical strategy that could transform coronavirus testing. Nature. 2020;583(7817):504-505. doi:10.1038/d41586-020-02053-6
- Cherif A, Grobe N, Wang X, Kotanko P. Simulation of Pool Testing to Identify Patients With Coronavirus Disease 2019 Under Conditions of Limited Test Availability. JAMA Netw Open. 2020;3(6):e2013075. doi:10.1001/jamanetworkopen.2020.13075
- Hogan CA, Sahoo MK, Pinsky BA. Sample Pooling as a Strategy to Detect Community Transmission of SARS-CoV-2. JAMA. 2020;323(19):1967–1969. doi:10.1001/jama.2020.5445
- Lohse S, Pfuhl T, Berkó-Göttel B, et al. Pooling of samples for testing for SARS-CoV-2 in asymptomatic people [published online ahead of print, 2020 Apr 28]. Lancet Infect Dis. 2020;S1473-3099(20)30362-5. doi:10.1016/S1473-3099(20)30362-5
- David Mastrianni, MD, Richard Falivena, DO, MPH, Timothy Brooks, MD, Brian McDermott, DO, Josenia Tan, MD, Richard Vandell, MS, MT(ASCP)SC, SH, Michael Holland, MD, Pooled Testing for SARS-CoV-2 in Hospitalized Patients. J Hosp Med. Published Online First July 20, 2020. DOI: 10.12788/jhm.3501
- Yates TA, Cooke GS and MacPherson P. Rational use of SARS-CoV-2 polymerase chain reaction tests within institutions caring for the vulnerable [version 1; peer review: 1 approved]. F1000Research 2020, 9:671 (https://doi.org/10.12688/f1000research.24872.1)
- Michael G. Becker, Tracy Taylor, Sandra Kiazyk, Dana R. Cabiles, Adrienne F.A. Meyers, Paul A. Sandstrom. Recommendations for sample pooling on the Cepheid GeneXpert® system using the Cepheid Xpert® Xpress SARS-CoV-2 assay. bioRxiv 2020.05.14.097287; doi: https://doi.org/10.1101/2020.05.14.097287