Purpose
Ophthalmology is the highest volume outpatient specialty within the NHS and the medicines used for medical retinal vascular conditions account for some of the highest cost and volume treatments used within secondary care. Due to increasing life expectancy and an ageing population, the NHS expects that demand for medical retinal vascular treatments will continue to increase in the future as more patients with eye disease are diagnosed and treated. Continually rising demand has also impacted ophthalmology outpatient services.
In response to these challenges, NHS England launched a comprehensive programme in 2024 to evaluate current and future treatment pathways within medical retinal services, with a focus on wet age-related macular degeneration, diabetic macular oedema, and retinal vein occlusion. The programme highlighted the need for a unified national treatment pathway, providing clear guidance on starting, switching, and stopping criteria to ensure patients receive optimal treatment at the appropriate stage. Additionally, a review of treatment data was conducted to identify the most cost-effective options while addressing ongoing capacity limitations.
The Commissioning framework for best value biological medicines sets out NHS England’s ambitions to establish a best value first approach, by accelerating and widening the adoption of best value biological medicines across the NHS – emphasising the importance of a collaborative approach.
The intent of these commissioning recommendations is to support the system to implement the national clinical pathways for medical retinal treatments, as well as the outcomes of the recent procurement exercise (with the framework commencing on 1 December 2025).
The pathways were developed with input from a national expert working group over 18 months. The group consisted of ophthalmologists, commissioners, pharmacists, patients and service managers across England, with most geographies represented. Recommendations made in this pathway were based on National Institute for Health and Care Excellence (NICE) guidance and clinical consensus, supported by published clinical trial and real-world NHS data. The pathways were also shared with local systems for consultation and feedback prior to publishing.
The treatment pathways were developed to support patients receiving the right treatments at the right point in the pathway and was guided by the following key objectives:
a. Reduction in unwarranted variation: reduce the number of patients that should be, but are not currently treated, and reduce the number of patients who are treated for whom treatment is inappropriate or ineffective. The aim is also to reduce variation across trusts in England on how they treated or managed patients.
b. Maintain clinical choice: clinicians will continue to determine, in discussion with their individual patients, which medical retinal vascular treatments are clinically appropriate for them and will be able to access all available treatments (in line with national guidance).
c. Make best use of NHS resources: to support the transformation of eyecare services (subject to the criteria specified in the relevant NICE technology appraisal guidance) clinicians should, in consultation with the patient, use the lowest cost treatment option where this is clinically acceptable.
Through consistently using the most clinically appropriate and cost-effective treatments and ensuring that patients receiving treatment are responding, we can drive efficiencies to support recovery and transformation in eyecare services. This will create capacity so that patients can be seen in a timely manner and avoid deterioration of vision. It also allows systems to treat patients earlier than NICE guidance, where this is commissioned and agreed locally.
These commissioning recommendations outline the best value treatment choices and, if followed, will generate financial savings which can be invested in NHS services.
Wet age-related macular degeneration (wet AMD)
Age-related macular degeneration is the leading cause of vision loss in the UK, impacting over 700,000 individuals. In recognition of the profound impact of sight preservation on patients’ quality of life, NHS England is committed to providing patients with high-quality, cost-effective care for wet AMD, whilst ensuring all those who might benefit from treatment receive it.
Commissioners are advised to use the Wet age-related macular degeneration pathway and calculator (The NHS Biosimilar Hub, Futures collaboration platform – registration required) to support with the strategic commissioning of a best value wet AMD pathway within their local area. Commissioners should assess the opportunity available to them from implementing the proposed pathway and work with their local trust and clinical teams to identify key savings opportunities using the calculator, whilst identifying any variation to the proposed approach with their local trusts.
Recommendations made in this pathway are based on NICE guidance and clinical consensus, supported by published clinical trial and real-world NHS data.
Treatment recommendations for wet AMD (appendix 1)
- Our analysis, combining both clinical trial data and real-world evidence, shows that adopting a treat-and-extend approach as standard – with aflibercept 2mg biosimilar – achieves the same patient outcomes at a lower cost. This makes it the best value option and should be used first line alongside ranibizumab biosimilar.
- This recommendation has looked at both medicines and activity costs. Whilst NICE deems all treatment options cost effective, this is based on the proviso that all patients respond to treatment a hundred percent and the NICE technology appraisals were not able to consider the role of biosimilars or identify the true associated activity costs.
- This treatment pathway offers a best value approach as a whole and outlines criteria that enable switching if patients do not respond fully to treatment or if they do not reach the expected dosing interval within a specific time interval. Adopting biosimilars helps unlock system-wide benefits allowing us to treat patients earlier (subject to local commissioning decisions). The savings generated from this ‘biosimilar first’ pathway frees up resources for reinvestment, for example to support efforts to reduce waiting lists in ophthalmology services.
Our modelling showed no significant difference in the number of injections between treatments, especially when treatment response is good. This is evidenced by real-world data from a sample of trusts. In other words, by using the treat and extend regimen, with aflibercept biosimilar as first line, this best value pathway will deliver the same clinical outcomes, cost significantly less, and likely have a minimal effect on capacity.
Diabetic macular oedema (DMO)
Diabetic macular oedema (DMO) is the most common cause for sight loss in people with diabetes. Almost 4.6 million people were diagnosed with diabetes in England in 2023/24, and the condition is more common in people of African-Caribbean and South Asian family origin than in those of European family origin. Approximately 7% of people with diabetes may have DMO in England, of whom 39% have clinically significant macular oedema (CSMO)*.
* Minassian et al (2012). Prevalence of diabetic macular oedema and related health and social care resource use in England. Br J Ophthal.96:345-349.
Effective management of diabetes and other risk factors may delay the onset and progression of DMO. This includes diet and lifestyle modification, blood pressure control and pharmacological treatments. For DMO specifically, the main treatments are anti-VEGF (anti-vascular endothelial growth factor treatments injected into the eye) therapy, steroid implants and laser photocoagulation. In recognition of the profound impact of sight preservation on patients’ quality of life, NHS England is committed to providing patients with high-quality, cost-effective care for DMO, whilst ensuring all those who might benefit from treatment receive it.
Commissioners are advised to use the Diabetic macular oedema pathway and calculator (The NHS Biosimilar Hub, Futures collaboration platform – registration required) to support with the strategic commissioning of a best value centre-involving diabetic macular oedema (CI-DMO) with visual impairment pathway within their local area. Commissioners should assess the opportunity available to them from implementing the proposed pathway and work with their local trust and clinical teams to identify key savings opportunities using the calculator, whilst identifying any variation to the proposed approach with their local trusts.
Recommendations made in this pathway are based on NICE guidance and clinical consensus, supported by published clinical trial and real-world NHS data.
- Treatment recommendations for CI-DMO (appendix 2). Our analysis, combining both clinical trial data and real-world evidence, shows that adopting a treat-and-extend approach as standard – with aflibercept 2mg biosimilar – achieves the same patient outcomes at a lower cost. This makes it the best value option and should be used first line alongside ranibizumab biosimilar.
- The number of appointments per patient over 3 years required is broadly similar across treatment options, with faricimab and aflibercept 8mg associated with slightly fewer appointments.
This treatment pathway offers a best value approach as a whole and outlines criteria that enable switching if patients don’t respond fully to treatment or if they don’t reach the expected dosing interval within a specific time interval. Adopting biosimilars helps unlock system-wide benefits and frees up resources to be reinvested into patient care.
Retinal Vein Occlusion (RVO)
Retinal vein occlusion (RVO) is the common cause of sudden painless reduction in vision in people over 60. It is the second most common cause of blindness. No prevalence or incidence data has been identified for England. In England between 2021 and 2022 there were 12,496 finished consultant episodes for RVOs, with 12,258 hospital admissions. The aims of current treatments are to preserve vision and prevent complications. Treatment is usually through anti-VEGF injections or steroid implants injected into the eye. The injections have to be repeated over a period of time to work effectively.
In recognition of the profound impact of sight preservation on patients’ quality of life, NHS England is committed to providing patients with high-quality, cost-effective care for RVO, whilst ensuring all those who might benefit from treatment receive it.
Commissioners are advised to use the Retinal vein occlusion pathway (The NHS Biosimilar Hub, Futures collaboration platform – registration required) to support with the strategic commissioning of a best value macular oedema secondary to RVO pathway within their local area. Commissioners should assess the opportunity available to them from implementing the proposed pathway and work with their local trust and clinical teams to identify key savings opportunities using the calculator, whilst identifying any variation to the proposed approach with their local trusts.
Recommendations made in this pathway are based on NICE guidance and clinical consensus, supported by published clinical trial and real-world NHS data.
No associated calculator was produced for RVO as systems can utilise the wet AMD calculator that has already been published.
Treatment recommendations for RVO (appendix 3)
- Our analysis, combining both clinical trial data and real-world evidence, shows that adopting a treat-and-extend approach as standard – with aflibercept 2mg biosimilar – achieves the same patient outcomes at a lower cost. This makes it the best value option and should be used first line alongside ranibizumab biosimilar. Aflibercept 8mg is not approved for use in RVO.
- This treatment pathway offers a best value approach as a whole and outlines criteria that enable switching if patients do not respond fully to treatment or if they do not reach the expected dosing interval within a specific time interval. Adopting biosimilars helps unlock system-wide benefits and frees up resources to be reinvested into patient care.
The main treatment goals for most patients
- Preservation of visual function (for example, best-corrected visual acuity [BCVA] improvement or stabilisation).
- Anatomical improvement from optical coherence tomography (OCT) (for example, lesion size, fluid in retina, haemorrhage) with no signs of disease activity.
- Manageable treatment burden for the patient.
However, it is recognised that not all patients can achieve complete disease remission despite frequent and timely dosing due to the progressive nature of the disease.
Recommendations for best practice
At treatment initiation, communicate with patients on all treatment possibilities at the outset. This would include:
- expected treatment outcomes and treatment burden with patients. Use real-world data to support communication, especially those with “poor” vision
- potential treatment changes throughout their journey, including the use of best value medicines when available
- potential for stopping treatment if there is no further clinical benefit with continued treatment.
See individual treatment pathways (appendices) for more detailed information on when to start treatment, when to switch and when to stop.
Table 1: summary of anti-VEGF and intravitreal corticosteroids via the NHS national framework agreement for England
|
Generic name |
Brand name |
Strength |
|
aflibercept biosimilar |
Afqlir® |
40 mg/mL solution for injection in pre-filled syringe |
|
aflibercept |
Eylea® |
40 mg/mL solution for injection in pre-filled syringe 114.3mg/mL solution for injection in pre-filled syringe |
|
aflibercept biosimilar |
Eydenzelt® |
40 mg/mL solution for injection in pre-filled syringe |
|
aflibercept biosimilar |
Vgenfli® |
40 mg/mL solution for injection in pre-filled syringe |
|
aflibercept biosimilar |
Yesafili® |
40 mg/mL solution for injection in pre-filled syringe |
|
bevacizumab |
Lytenava® |
25mg/mL solution for injection in a vial |
|
brolucizumab |
Beovu® |
120 mg/mL for injection in pre-filled syringe |
|
dexamethasone |
Ozurdex ® |
700microgram intravitreal implant in applicator |
|
faricimab |
Vabysmo® |
120mg/mL solution for injection in a vial 120mg/mL solution for injection in pre-filled syringe |
|
fluocinolone acetonide |
Iluvien ® |
190microgram intravitreal implant in applicator |
|
ranibizumab biosimilar |
Byooviz® |
10mg/mL solution for injection in a vial |
|
ranibizumab |
Lucentis® |
10mg/mL solution for injection in a vial 10mg/mL solution for injection in pre-filled syringe |
|
ranibizumab biosimilar |
Ongavia® |
10mg/mL solution for injection in a vial |
|
ranibizumab biosimilar |
Rimmyrah® |
10mg/mL solution for injection in a vial |
Appendix 1: treatment algorithm for wet AMD
Refer to notes in the Wet age-related macular degeneration pathway (The NHS Biosimilar Hub, Futures collaboration platform – registration required).
If more than 1 treatment is suitable, use the least expensive treatment. Use best value brand available locally.
This guideline is based on treat-and-extend protocol, which is the preferred regime for most patients and services. It is recognised that some patients may benefit from regular treatment intervals to aid adherence.
This flow diagram is not fully accessible. If you require the information in an alternative format please email england.medsvaluepharmacists@nhs.net

Table 2. Drug dosing details according to summary of product characteristics (SPC) recommendations
|
Drug |
Posology post-loading – no disease activity |
Treat-and-extend dose increment intervals – disease activity |
Maximum treatment intervals |
Minimum dose intervals | |
|
|
| ||||
|
First choice | |||||
|
Ranibizumab biosimilar |
Treat-and-extend
|
Continue monthly |
2 weeks
|
12 weeks |
4 weeks |
|
Aflibercept 2mg
Biosimilar once available |
Treat-and-extend
|
Continue 2-monthly |
2-4 weeks |
16 weeks |
4 weeks |
|
Second choice | |||||
|
Aflibercept 8mg |
Treat-and-extend
|
Clinical decision |
Not specified |
16 weeks, can be further extended to 24 weeks |
8 weeks* |
|
Faricimab |
Treat-and-extend
|
Continue 8-12 weekly |
4 weeks |
16 weeks |
4 weeks** |
|
Third choice | |||||
|
Bevacizumab |
Treat-and-extend |
Continue monthly |
Not specified |
12 weeks |
4 weeks |
|
Brolucizumab |
Every 3 months
|
Every 2 months |
Not specified |
12 weeks |
8 weeks |
Off-license dosing details
*Aflibercept 8mg off-license dosing: max 4 weekly for 3 consecutive doses were used in studies.
**Faricimab off-license dosing: 3 weekly was used in studies to allow flexibility of dose scheduling.
The safety and efficacy of off-license dosing has not been evaluated. Therefore, NHS England do not recommend routine commissioning of off-license dosing.
Figure 1. indicative combined costs (drug and activity) based on average number of doses from NHS England modelling and real-world NHS data at the time of writing
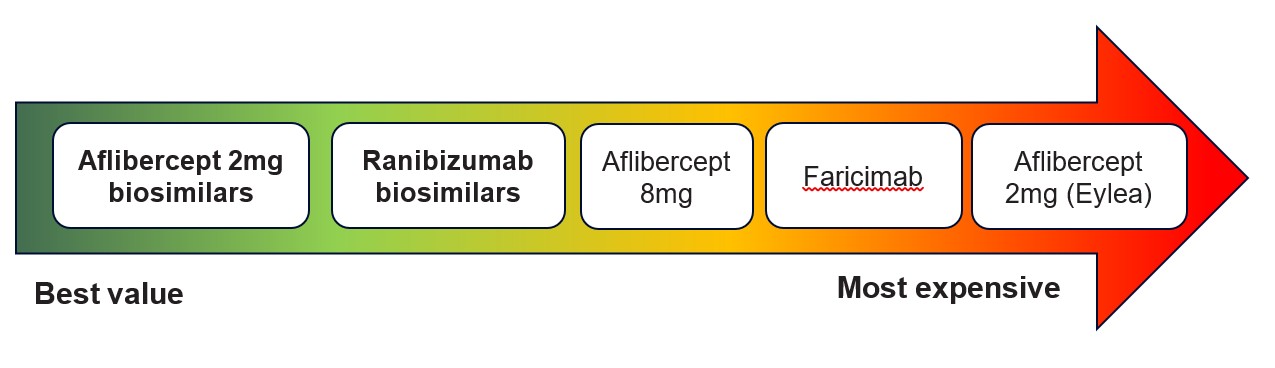
Figure 1 shows a list of combined costs (drugs and activity) from best value (top of list) to most expensive (bottom of list):
- Aflibercept 2mg biosimilars
- Ranibizumab biosimilars
- Aflibercept 8mg
- Faricimab
- Aflibercept 2mg (Eylea)
Appendix 2: Treatment algorithm for centre-involving diabetic macular oedema with visual impairment
Refer to notes in Diabetic macular oedema pathway (The NHS Biosimilar Hub, Futures collaboration platform – registration required).
If more than 1 treatment is suitable, use the best value treatment and best value brand available locally through national procurement framework. Prescribe by brand.
This flow diagram is not fully accessible. If you require the information in an alternative format please email england.medsvaluepharmacists@nhs.net
Figure 1. Recommended anti-VEGF efficacy and treatment burden check time points
This flow diagram is not fully accessible. If you require the information in an alternative format please email england.medsvaluepharmacists@nhs.net

Table 5. anti-VEGF dosing details according to SPC
General information for all anti-VEGFs
Based on the physician’s judgement of visual and/or anatomic outcomes, the treatment intervals can be increased to maintain stable visual and/or anatomic outcomes.
If visual and/or anatomic outcomes deteriorate, the treatment interval should be shortened accordingly.
|
Anti-VEGF |
Loading dose |
Maintenance dose (post-loading, with disease activity) |
Treat-and-extend dose increment intervals (maximum treatment intervals) |
Minimum dose intervals |
|
First choice | ||||
|
Aflibercept 2mg (TA346) (switch to biosimilars once available) |
2mg monthly for 5 months |
1 injection every 2 months |
2 weeks (up to 1 injection/ 16 weeks) |
4 weeks
|
|
Ranibizumab biosimilar (TA274)
|
0.5mg every 4 weeks for 3 months or more until maximum visual acuity achieved |
4 weekly |
Up to 4 weeks (no specific maximum treatment intervals specified in SPC) |
4 weeks
|
|
Second choice | ||||
|
Aflibercept 8mg
|
8mg monthly for 3 months |
1 injection every 2 months |
Not specified (up to 1 injection/16 weeks, can be further extended to 1 injection/24 weeks) |
8 weeks*
|
|
Faricimab (TA799)
|
6mg every 4 weeks for 3 doses |
1 injection every 8 to 16 weeks |
Up to 4 weeks (up to 1 injection/16 weeks) |
4 weeks** |
|
Third choice | ||||
|
Brolucizumab (TA820) |
6mg every 6 weeks for 5 doses |
Without disease activity: 1 injection every 12 weeks With disease activity 1 injection every 8 weeks |
Not specified (up to 1 injection/16 weeks). Extension should only happen after 12 months of treatment |
8 weeks
|
Off-license dosing details
*Aflibercept 8mg off-license dosing: max 4 weekly for 3 consecutive doses were used in studies.
**Faricimab off-license dosing: 3 weekly was used in studies to allow flexibility of dose scheduling.10,11
The safety and efficacy of off-license dosing has not been evaluated. Therefore, NHS England do not recommend routine commissioning of off-license dosing.
For patients with baseline Early Treatment Diabetic Retinopathy Study (ETDRS) < 69 letters, Protocol T study demonstrated efficacy hierarchy for the following drugs: aflibercept (most efficacious) > ranibizumab > bevacizumab (least efficacious).1210 There is lack of evidence which directly compares the efficacy hierarchy for all anti-VEGFs which have UK marketing authorisation.
Figure 2. Indicative anti-VEGF combined costs (drug and activity) based on average number of doses from NHS England modelling and real-world NHS data at the time of writing

Figure 2 shows a list of indicative anti-VEGF combined costs (drugs and activity) from best value (top of list) to most expensive (bottom of list):
- Aflibercept 2mg biosimilars
- Ranibizumab biosimilars
- Aflibercept 8mg
- Faricimab
- Aflibercept 2mg (Eylea)
Steroid implant dosing details according to SPC
Preferred choice: dexamethasone (TA824)
Dose
700 micrograms (1 implant) intravitreally into the affected eye.
Manufacturer recommends retreatment may be performed after approximately 6 months if the patient experiences decreased vision and/or an increase in retinal thickness, secondary to recurrent or worsening diabetic macular oedema.
In clinical practice, therapeutic effect can last for 4 months and may require earlier retreatment (off-label). The expert working group is unable to clinically define this cohort.
Other notes
Administration to both eyes concurrently is not recommended. Common clinical practice is to administer 1 eye at a time for the first injection to review for response and side effects. If no concerns and clinically appropriate, clinicians may choose to administer both eyes at the same time.
Manufacturer reports no experience of the efficacy or safety of repeat administrations in diabetic macular oedema (DMO) beyond 7 implants. Clinicians report no concerns and the number of patients requiring this is small.
Subject to local commissioning, it is recommended to commission up to 3 implants per year (or treatment every 4 months) [off-label] until the patient meets discontinuation criteria (see note 5 and note 9).
Fluocinolone (TA953)
Dose
190 micrograms (one implant) intravitreally into the affected eye.
An additional implant may be administered after 12 months if the patient experiences decreased vision or an increase in retinal thickness secondary to recurrent or worsening diabetic macular oedema.
Other notes
Retreatments should not be administered unless the potential benefits outweigh the risks.
Only patients who have been insufficiently responsive to prior treatment with laser photocoagulation or other available therapies for diabetic macular oedema should be treated with fluocinolone.
The safety and efficacy of fluocinolone administered to both eyes concurrently has not been studied. It is recommended that an implant is not administered to both eyes at the same visit.
Subject to local commissioning, rescue treatments (for example, anti-VEGFs, laser, dexamethasone implant) are recommended whilst on fluocinolone implant (see note 5). This affects approximately 40% according to the Fluocinolone Acetonide for Diabetic Macular Edema (FAME) study and Intelligent Research In Sight (IRIS) registry study.
Subject to local commissioning, up to 2 implants per eye is recommended to be commissioned.
Indicative combined costs (drug and activity) of anti-VEGFs and steroid implants based on average number of doses from NHS England modelling and real-world NHS data at the time of writing
List of combined drugs from best value (top of list) to most expensive (bottom of list)
- Aflibercept 2mg biosimilars (best value)
- Ranibizumab biosimilars (best value)
- Aflibercept 8mg
- Faricimab
- Dexamethasone implant (6 monthly/ 2 implants per year)
- Fluocinolone implant (1 implant over 3 years)
- Aflibercept 2mg (Eylea)
- Dexamethasone implant (4 monthly/ 3 implants per year)
- Fluocinolone implant (2 implants over 3 years)
Note: the cost of rescue treatments needed for fluocinolone implant is not included in the model.
Table 7. estimated loss of exclusivity (LoE)
| Drug | Estimated LoE |
| Ranibizumab | July 2022 |
| Aflibercept 2mg | End of November 2025 |
| Aflibercept 8mg | 2039 |
| Faricimab | 2037 |
| Brolucizumab | 2034 |
| Dexamethasone implant | 2026 (no planned generics launch at the time of writing) |
| Fluocinolone acetonide implant | Expired-pending on regulatory approval of generics |
Note: patents can be liable to legal challenge and dates may change. Some medicines may be subject to additional patents on, for example, therapeutic use or device.
Appendix 3: Treatment algorithm for macular oedema secondary to RVO
Refer to notes in Retinal vein occlusion pathway (The NHS Biosimilar Hub, Futures collaboration platform – registration required)
Choice of treatment between anti-VEGFs or steroid implants will need to consider risk of intraocular pressure (IOP), cardiovascular risk, cataract formation and injection frequency.
If more than 1 treatment is suitable, use the best value treatment based on the overall costs (drug and service delivery). Prescribe by brand and use best value brand available locally.
This flow diagram is not fully accessible. If you require the information in an alternative format please email england.medsvaluepharmacists@nhs.net

Figure 1: schematic illustration of anti-VEGF monitoring for RVO
This flow diagram is not fully accessible. If you require the information in an alternative format please email england.medsvaluepharmacists@nhs.net

Table 2: anti-VEGF dosing details according to SPC recommendations
General information for all anti-VEGFs
Consider switch to steroid implant if recent (within 6 months) cardiovascular events, unable to comply to injection frequency with anti-VEGFs (see also note 6)
Aflibercept 8mg is not licensed for use in RVO.
|
Anti-VEGF |
Cost tier |
Initiation phase |
Maintenance phase |
Treat-and-extend (T&E) dose increment intervals |
Minimum T&E dose intervals |
|
First line | |||||
|
Aflibercept 2mg (TA305, TA409)
|
£££ – most expensive
|
1 injection per month until maximum visual acuity is achieved and/or there are no signs of disease activity.
3 or more consecutive, monthly injections may be needed. In practice this is normally 3 to 6 injections. |
If there is no change in disease activity, maintenance therapy could be according to a treat-and-extend regimen or a when required (PRN) treatment regimen
Treat-and-extend regimen: Based on the physician’s judgement of visual and/or anatomic outcomes, the treatment intervals can be increased to maintain stable visual and/or anatomic outcomes. If visual and/or anatomic outcomes deteriorate (CMT worsens compared to baseline), the treatment interval should be shortened accordingly.
In CRVO, RCOphth guidance recommends the following: “once this interval to recurrence is identified, it is advisable to maintain on this interval for a 6-month period before extending again”. |
Not specified in SPC, insufficient data to conclude maximum treatment intervals |
4 weeks |
|
Biosimilars once available |
£ – cheapest |
1 injection per month until maximum visual acuity is achieved and/or there are no signs of disease activity.
3 or more consecutive, monthly injections may be needed. In practice this is normally 3 to 6 injections. |
If there is no change in disease activity, maintenance therapy could be according to a treat-and-extend regimen or a PRN treatment regimen
Treat-and-extend regimen: Based on the physician’s judgement of visual and/or anatomic outcomes, the treatment intervals can be increased to maintain stable visual and/or anatomic outcomes. If visual and/or anatomic outcomes deteriorate (CMT worsens compared to baseline), the treatment interval should be shortened accordingly.
In CRVO, RCOphth guidance recommends the following: “once this interval to recurrence is identified, it is advisable to maintain on this interval for a 6-month period before extending again”. |
Not specified in SPC, insufficient data to conclude maximum treatment intervals |
4 weeks |
|
Ranibizumab 0.5mg biosimilars (TA283)
|
£ – cheapest |
1 injection per month until maximum visual acuity is achieved and/or there are no signs of disease activity.
3 or more consecutive, monthly injections may be needed. In practice this is normally 3 to 6 injections. |
If there is no change in disease activity, maintenance therapy could be according to a treat-and-extend regimen or a PRN treatment regimen
Treat-and-extend regimen: Based on the physician’s judgement of visual and/or anatomic outcomes, the treatment intervals can be increased to maintain stable visual and/or anatomic outcomes. If visual and/or anatomic outcomes deteriorate (CMT worsens compared to baseline), the treatment interval should be shortened accordingly.
In CRVO, RCOphth guidance recommends the following: “once this interval to recurrence is identified, it is advisable to maintain on this interval for a 6-month period before extending again”. |
Not specified in SPC, insufficient data to conclude maximum treatment intervals |
4 weeks |
|
Second line |
|
|
| ||
|
Faricimab 6mg (TA1004)
|
££ – mid-range price |
1 injection per month until maximum visual acuity is achieved and/or there are no signs of disease activity.
3 or more consecutive, monthly injections may be needed. In practice this is normally 3 to 6 injections. |
If there is no change in disease activity, maintenance therapy could be according to a treat-and-extend regimen or a PRN treatment regimen
Treat-and-extend regimen: Based on the physician’s judgement of visual and/or anatomic outcomes, the treatment intervals can be increased to maintain stable visual and/or anatomic outcomes. If visual and/or anatomic outcomes deteriorate (CMT worsens compared to baseline), the treatment interval should be shortened accordingly.
In CRVO, RCOphth guidance recommends the following: “once this interval to recurrence is identified, it is advisable to maintain on this interval for a 6-month period before extending again”. |
Up to 4 weeks. Max treatment interval 16 weeks (no data for > 16 weeks intervals) |
4 weeks*
|
Cost tier per annum (drug and activity) based on number of doses expected per annum
£ (cheapest) to £££ (most expensive) from NHS England modelling (see section below) at the time of writing.
*Faricimab off-license dosing: 3 weekly was used in studies to allow flexibility of dose scheduling.
The safety and efficacy of off-license dosing has not been evaluated. Therefore, NHS England do not recommend routine commissioning of off-license dosing for faricimab.
Steroid implant dosing details according to SPC recommendations
Dexamethasone intravitreal implant (TA824)
2 injections per year ££
3 injections per year £££
Dose
700 micrograms (1 implant) intravitreally into the affected eye.
Repeat doses should be considered when a patient experiences a response to treatment followed subsequently by a loss in visual acuity and in the physician’s opinion may benefit from retreatment without being exposed to significant risk.
Patients who experience and retain improved vision should not be retreated.
Patients who experience deterioration in vision, which is not slowed by dexamethasone should not be retreated (consider stopping permanently, see (see note 8).
Other notes
Administration to both eyes concurrently is not recommended.
Royal College of Opthalmolgy (RCOphth) guidance states that if this is used as first line, re-treatment at every 4 (off-label) to 6 monthly (licensed) intervals may be required until visual stability is obtained. The occasional patient may require treatment at 3 months (off-label). However, repeated and frequent treatments will increase the risk of adverse events and these should be discussed with the patient.
NICE TA only included 6-monthly intervals in their economic assessment.
Subject to local commissioning, it is recommended to commission up to 3 implants per year (or treatment every 4 months) [off-label] until the patient meets discontinuation criteria [off-label] (see note 3 and note 8).
Table 4: estimated loss of exclusivity (LoE)
|
Drug |
LoE |
|
Ranibizumab |
July 2022 |
|
Aflibercept 2mg |
End of November 2025 (8mg not licensed in RVO) |
|
Faricimab |
2037 |
|
Dexamethasone implant |
2026 (no planned generics launch in UK) |
Note: patents can be liable to legal challenge and dates may change. Some medicines may be subject to additional patents on, for example, therapeutic use or device.
Publication reference: PRN00087

