Applicability
This NETB applies to all healthcare spaces with ventilation requirements.
Objective
To provide additional technical guidance and standards on the use of UVC devices for air cleaning in healthcare spaces.
Status
The document represents advice for consideration by all NHS bodies. It is to be read alongside Health Technical Memorandum 03-01 Specialised Ventilation for Healthcare Premises (HTM 03-01).
Point of contact/feedback
Point of contact for any queries: england.estatesandfacilities@nhs.net
Executive summary
Ventilation* is a key line of defence for infection control in the healthcare environment. Its design and operation are described in Health Technical Memorandum (HTM-03-01). The current focus on ventilation has highlighted areas of high risk due to poorly performing and inadequate ventilation in hospitals and other healthcare settings due to age, condition of air handling plant, lack of maintenance, challenges with effective use of natural ventilation or other creates areas of high risk. It is therefore important to bring these facilities up to the minimum specification of current standards, particularly recognising the challenges of COVID-19 and other respiratory infections.
Ultraviolet (UVC) air cleaners (also known as air scrubbers) using ultraviolet light are one option for improving and upgrading ventilation. The installation of a UVC air cleaner can reduce the risk of airborne transmission.
This document has been written as an interim specification to set the basic standard required for UVC devices to be utilised in healthcare and patient related settings. This edition is primarily aimed at portable and semi fixed (wall-mounted) devices. The series will extend to in-duct and upper room devices in future iterations. Devices relying on HEPA filters or similar filter-based technology can have similar benefits to UVC devices but are not considered in this document. The potential of air scrubbers employing UVC or HEPA technology is the subject of a rapid review (September 2022).
*Ventilation is the process by which ‘fresh’ air (normally outdoor air) is intentionally provided to a space and stale air is removed. This may be achieved by mechanical systems using ducts and fans, or natural ventilation most commonly provided through opening windows. The local redistribution of air may also be construed as ventilation.
1. Introduction
Ventilation is a critical feature in the control of airborne infection. However, the emergence of SARS-CoV-2 as a highly contagious virus has demanded new and innovative solutions to safeguard patients, staff and visitors. Health Technical Memorandum 03-01 Specialised Ventilation for Healthcare Premises (HTM-03-01) is a robust standard for ventilation of higher risk clinical spaces based on high air change rates using outdoor air to continually flush indoor spaces. The emergence of COVID-19 has shown that greater attention must be paid to the removal or deactivation of airborne pathogens in areas where ventilation rates are lower.
The focus on ventilation has also highlighted areas of high risk due to poorly performing and inadequate ventilation, particularly in older hospitals and other healthcare settings such as primary care and dental, which increase risks of infection spread viz nosocomial infections.
In cases, where current ventilation does not meet HTM-03-01 standards, this may be due to age, condition of air handling plant, lack of maintenance or other design or operational issues. In the case of naturally ventilated spaces, there is a reliance on staff or patients opening windows. Weather conditions, external noise and air pollution and restricted window openings for safety affect the ability to open windows and means that ventilation in some settings can fall below recommended rates.
UVC air cleaners using ultraviolet light are one option for improving and upgrading ventilation. The correct installation and operation of a UVC air cleaner can effectively reduce the risk of airborne transmission.
NHS trusts are under pressure to improve ventilation and are considering options including UVC air cleaning. This standard will assist trusts in selecting and implementing good quality, reliable equipment.
There is substantial evidence from laboratory studies and real-world settings that UVC is an effective technology for reducing airborne pathogens within room air and HVAC systems. A number of trial ‘case studies’ have been carried out which indicate that measured levels of microorganisms in air are greatly reduced and infection rates have decreased.
These trials have also shown that UVC within HVAC systems safely allows some levels of air recirculation and can achieve substantial energy reductions compared to the normal 100% fresh air approach set out in HTM-03-01. For example, a scheme with 50% fresh air and 50% recirculated air would reduce heat demand by 50%. However, care must be taken to ensure sufficient fresh air changes are provided for the dilution of medical gases and noxious odours, and the maintenance of appropriate oxygen and carbon dioxide levels.
This document aims to serve as interim guidance and regulatory reference point for the design and correctly engineered deployment of germicidal UVC devices in real-world settings with regard to effectivity and safety.
2. UVC germicidal effects
There are a wide range of UVC devices which aim to inactivate microorganisms in the air and/or on surfaces. This document focuses on contained UVC devices which can be positioned locally within a room or within an HVAC duct. These devices usually require fan assisted circulation to introduce the room air into the device, expose it to ultraviolet light and then to reintroduce the processed air into the room. Therefore, aerodynamics internal to the device together with the lamp specification determines the air and microbial particle UVC exposure time and hence the radiation dose.
These devices are known as active UVC air cleaning devices. Not considered in this document are passive UVC devices, aka upper room devices, which rely on the natural air currents within rooms.
An important consideration regards the flow of the air which is induced, processed and distributed by the device external to the device itself. The design and placement of the device should promote efficient air circulation in the room space and avoid short-circuiting of air circulation relative to furniture, obstructions, and occupancy.
The ultraviolet-C (UVC) spectrum lies in the interval [200…280] nm. UVC irradiation as a means of microbial inactivation has been used for over 100 years in multiple sectors including medical, scientific, water disinfection, manufacturing and agricultural.
UVC germicidal activity inactivates microorganisms rendering them unable to replicate. Most commonly, germicidal activity is generated by mercury ionisation lamps with the major spectral line at 254 nm wavelength. This is sometimes also known as germicidal ultraviolet (GUV) or ultraviolet germicidal irradiation (UVGI). This standard uses the term UVC.
Recent studies suggest that devices based on far-UV (222 nm wavelength) may also be effective; however, these are not covered here.
The photo-toxicity risks associated with UVC is universally recognised. The design, specification and implementation of germicidal UVC solutions currently lacks rigorous governance and the requirement for regulatory change is recognised. The purpose of this standard therefore is to establish the key criteria for successful and reliable long-term application of UVC air cleaning while avoiding the potential safety hazards and operational pitfalls, particularly when equipment is used in spaces occupied by non-technical people.
3. Applications
This standard covers the types of UVC air cleaners used as standalone or in-duct units where the principal active element is UVC at the nominal wavelength of 254 nm.
In rooms without natural or mechanical ventilation, or where the ventilation falls short of local requirements or regulatory advice, auxiliary devices may be deployed to enhance the effective air changes. The installation of UVC air cleaners can be considered to contribute additional ‘equivalent’ air changes (eACH). For example, a treatment room with only 2 ACH could achieve the equivalent of 10 ACH by installing a UVC unit which recirculated and cleaned the equivalent of 8 ACH (eACH) for the microorganisms of concern. Hence, to meet the requirements that comply with HTM-03-01, the number of devices required will be dictated by the existing background levels of ventilation.
In-duct HVAC systems
In buildings with existing HVAC systems which have recirculation of air, it can be effective to install UVC lamps directly into the ducts, placing them downstream of pre-existing particulate filters. This allows for the treatment of all rooms in the building covered by the HVAC system or within branch ducts serving various zones and the rooms within those zones.
Due to the lamps being contained within the ducts, the risk of direct exposure to UVC is low. However, maintenance can be carried out safely shut-down interlocks should be fitted and hazard notices compliant with BS EN ISO 7010 prominently displayed.
Standalone devices
Standalone devices maybe portable (floor-standing) or fixed (wall- or ceiling-mounted).
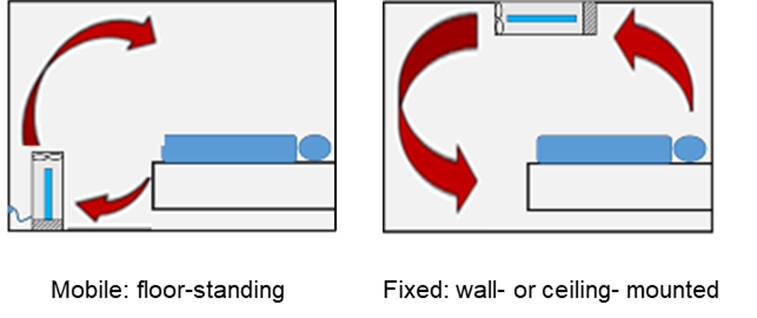
Figure: Representation of air flows with respect to a recumbent patient in a regular room for 2 device locations. i. mobile: floor-standing; ii. fixed: wall- or ceiling-mounted.
254 nm devices covered in this standard
- In-duct UVC: UVC lamps are installed directly into the HVAC system or are contained within a locally installed ventilation device which is connected into the HVAC system, similar to a fan-coil unit. Devices may use the fans and filters within the existing HVAC system or, in some cases, may have local fans and filters to provide the recirculation. Significant modelling and design are required to implement such systems.
- Floor standing UVC ‘mobile’ devices: UVC lamps are contained within a standalone floor mounted device that can be positioned at any suitable location in a room. These devices provide local air cleaning within a room and are plugged into a standard electrical socket so do not require any installation. The device contains lamps, dust filters and a fan to draw room air through the device. Devices are portable and so can be easily moved.
- Fixed UVC devices – wall or ceiling mounted: Similar to floor standing units but fixed to a wall or ceiling.
These devices will normally be permanently wired into the room electrical system rather than plugged into a wall socket.
UVC devices not covered in this standard
- Decontamination UVC devices: High intensity open-field UVC devices that are designed for periodic surface decontamination in unoccupied spaces. These devices are sometimes known as UVC robots.
- Upper-room UVC devices: UVC devices which utilise an open UV field within the room above the heads of occupants. These are passive devices which rely on the general circulation of room air and are sometimes assisted by ceiling fans.
- Devices based on other parts of the UV spectrum: The devices covered in this standard are based on 254 nm wavelength lamps. There are a number of other UV technologies including Far UV (222 nm) which has early data showing it is likely to be effective.
- Devices that incorporate other technologies alongside UVC: There are a number of devices which use UVC alongside other technologies such as titanium dioxide catalysts or ionisers. These devices often emit by-products into the room, either intentionally or deliberately. The health impacts of any emissions must be carefully considered.
4. Safety
4.1 Accidental exposure
Safety is of paramount importance when working with UVC devices. Direct exposure to UVC light can cause damage to the skin and eyes.
The manufacturer of a germicidal UVC device should provide assurance in the device specification that the maximum UV (total) irradiance at 20 cm distance from any part surface of the device is ≤1 mW.m2 (noting that this is based on an accumulated exposure of 8 hours). Exposure limits to UVC are specified in the directive Control of Artificial Optical Radiation at Work Regulations (AOR) 2010.
Fail-safe systems are required to prevent lamps from operating when the cover of the device is removed.
4.2 Wider safety considerations
Care needs to be taken during maintenance and in operation that lamps are not broken. Appropriate safety protocols need to be in place to minimise risk of exposure to mercury vapour where devices contain mercury based lamps.
As electrical devices, UVC devices must comply with the Low Voltage Designated Standards (Electrical Equipment (Safety) Regulations 2016).
Manufacturers should be aware that wiring and other components are liable to degradation under UV radiation.
5. Engineering implementation
5.1 Regulatory and standards compliance
Standards are an integral part of product design and development and are important in medical applications. The Low Voltage Directive (section 5.1.2) should be followed implicitly as a minimum. There are other standards and regulations which apply when using UVC air cleaning devices.
IEC 60601 is a series of technical standards which apply to medical electrical equipment and medical electrical systems for basic safety and essential performance. The basic scope of IEC 60601 -1 is the safety of patients and users. While compliance to IEC 60601-1 is not mandated in this standard, the design of standalone germicidal UVC devices should follow the principles of the 6061 standard to ensure risks to patient and user safety within a medical environment are recognised and mitigated (section 5.1.2)
5.1.1 CE and UKCA marking
CE and UKCA marking are standards that appear on products traded on the extended single market in the European and UK economic areas. The marking signifies that the product has been assessed to meet high health, safety and environmental requirements.
- Selling products in Europe:
- use of the CE-mark declares that the product meets the legal requirements for sale throughout the European Union.
- Selling products in the UK:
- the UKCA-mark is the product marking used for products being placed on the market in Great Britain (England, Scotland and Wales)
- the UKCA-mark applies to most products previously subject to the CE- marking. The technical requirements (sometimes referred to as ‘essential requirements’) must be met.
5.1.2 Electrical safety
- Compliance with the Low Voltage Directive is mandated implicitly.
- Compliance with the IEC 60601‑1 standard is explicitly mandated.
- Class I (exposed metal components connected to earth):
- protective earth continuity <0.2 MΩ.
- insulation tests: ≥50 MΩ
- earth leakage: ≤5 mA in normal condition (NC), ≤10 mA in SFC (single fault condition)
- enclosure leakage current: ≤1 mA in NC, ≤0.5 mA in SFC
- Class II (double-insulated enclosure):
- insulation tests: ≥50 MΩ.
- enclosure leakage current: ≤0.1 mA in NC, ≤0.5 mA in SFC
Class III devices are not recommended.
5.1.3 Electrical wiring
Electrical wiring should be in accordance with IET Regulations BS 7671:2018 Requirements for Electrical Installations.
Electrical components which are contained within a UVC device must be selected appropriately. Wiring and connectors should not be exposed to direct high intensity UV light. However, where exposure is unavoidable, secondary UV-resistant sheath should be employed. Exposed cables, particularly any with PVC coverings, will deteriorate due to the effect of UVC light.
5.1.4 Optical radiation safety
Safety is of paramount importance when working with UVC devices. Direct exposure to UVC light can cause damage to the skin and eyes.
The manufacturer of a germicidal UVC device should provide assurance in the device specification that the maximum UV (total) irradiance at 20 cm distance from any part surface of the device is ≤1 mW.m2 (noting that this is based on an accumulated exposure of 8 hours). Exposure limits to UVC are specified in the directive Control of Artificial Optical Radiation at Work Regulations (AOR) 2010.
Fail-safe systems are required to prevent lamps from operating when the cover of the device is used.
5.2 Ozone hazard
Ozone, an allotrope of oxygen, can be produced when oxygen is exposed to UVC with a wavelength below 240 nm. Ozone above occupational exposure limits (UK Workplace Exposure Limit (WEL) of 0.2 ppm (15 minute reference period)) is harmful to human health and can affect the respiratory, cardiovascular and central nervous system. Ozone can also cause degradation of certain materials, which can lead to fire hazards.
Manufacturers shall provide assurance that devices do not produce ozone which contributes to room levels in excess of the WEL.
6. Engineering design, specification and performance validation
6.1 Characteristic specification (characteristic verification)
The manufacturer should provide a 10 mm diameter access port to the reaction chamber. This will enable the point measurement of air velocity and point measurement of UVC irradiance to provide assurance that the device is operating to the specification cited by the manufacturer under ‘verification’. It is expected that this facility will be used during the annual maintenance check by the designated competent persons (section 7).
6.2 Bio-validation
The microbial inactivation rate for a UVC device, and hence the equivalent air change rate it provides, depends on the microorganism and the temperature and humidity. The manufacturer should provide evidence of the germicidal effectivity of their device at a given air flow (see above) and under given environmental conditions. At the present time, the preferred method of bio-validation (the Liverpool Biovalidation Protocol for the real-world evaluation of UVC-based air purifiers (NHS England Supply Chain)) uses Micrococcus luteus as the bacterial challenge under ambient environmental conditions of 23 C and a relative humidity of 50%. If an alternative protocol is employed, equivalence must be evidenced with reference to k, the UVC susceptibility constant for the particular microorganism (k, inactivation rate constant (susceptibility rate) [cm2.mJ-2]).
Where devices are used in settings where particular pathogens are likely to pose hazard, it is important to ensure that the susceptibility of the pathogen to UVC is taken into account when selecting a device.
6.3 Lamp guidance
At the time of publication, the most common source of UVC radiation is the mercury-vapour lamp (aka the mercury gas-discharge lamp). These devices are designed to emit at the wavelength 254 nm. While other technologies are available, eg. light emitting diodes (LEDs) and amalgam-mercury based discharge tubes, they are not considered here. Lamps should have anti-static surface coatings to minimise the build-up of surface contamination.
6.3.1 Effective life span
Lamp lifespan should be optimised to minimise replacement times and allow for a straight-forward replacement schedule.
Lamps should have an effective operational life of no less than one year (circa 8,800 hours for 24/7 active operational life) before they need replacing. Typically, the optical efficiency of a mercury-vapour lamp will decrease by 20% over its effective life span.
6.3.2 Operating conditions
The efficiency of a mercury-vapour lamp is affected by ambient temperature. Manufacturers should provide assurance that devices deliver their germicidal potency, as claimed, over an environmental operating temperature range of [10 …35] C.
6.3.3 Lamp failure indication
An alarm (visual and/or audible) should be implemented to notify of lamp failure.
6.4 Noise considerations
Devices in normal operation in occupied areas should operate at a sound level of ≤50 dB measured at 3 m (dB3m). Exceptionally, for operation at boost, such that might be used to purge a room with controlled occupancy, the sound level should not exceed 60dB3m
Noise is a particular consideration when devices are used in rooms where patients are sleeping, and lower sound levels than stated here may be required depending on local environmental conditions. Further guidance on wider considerations around acoustics in healthcare is given in HTM-08-01.
7. Competent persons
In the present context, competent persons are recognised as individuals with professional expertise in one or more of the following areas in the healthcare setting: the design of UVC systems, the technical maintenance of UVC devices and systems, and the implementation of air sanitization schemes employing germicidal UVC.
Further, competent persons with particular expertise in infection prevention and control are essential to identify the relevant target microorganisms that UVC devices will need to mitigate.
8. Engineering and operational considerations
8.1 Hazard, risk and operational delivery
A ventilation design incorporating UVC-based air cleaners will require a hazard and operational study (HAZOP). This process will be convened by the Ventilation Safety Group (a group of individuals with recognised expertise in the design and operation of ventilation devices and systems responsible for the governance of the device deployments, as defined in HTM 03-01) which will include competent persons (section 7) including representation from infection and prevention control, nursing and estates management and/ or clinical engineering.
8.2 Conventional HVAC filters
Filters should be included into UVC systems to protect the UV lamps from dust build-up such that UV fluence is not compromised. Some devices may also contain carbon filters to mitigate odour and VOCs. In normal operation, the replacement period for such filters should not be less than one year. In exceptional circumstances, such as operation in areas with high levels of large particulate contamination, more regular replacement may be required to ensure air flow is not restricted. Local Standard Operating Procedures (SOPs) should be applied.
8.3 Ventilation effectiveness
The Ventilation Safety Group (HTM 03-01) will consider air flow strategies which achieve the most effective ventilation of occupied spaces. This requires that all factors such as air flow rate, mixing and distribution, dilution, thermal buoyancy and the impact of occupant movements must be considered.
8.3.1 Computational fluid dynamics (CFD) modelling of air movement
CFD modelling can be a useful tool to assist ventilation engineers to assess airflow patterns in the rooms where UVC devices are to be used and to identify the optimal locations to place devices. CFD modelling requires specialist knowledge, any simulations should be carried out by a competent person. CFD simulations can illustrate typical airflow patterns but may not be able to capture all of the fluctuations that occur in real environments, particularly those that are naturally ventilated.
8.4 Installation
The installation of any UVC air scrubbing devices should comply with all local building and electrical guidance. Advice should be sought from competent persons (section 7) including representation from infection and prevention control, nursing and estates management and/ or clinical engineering.
When positioning portable units engineers should consider the manufacturer’s recommendations around the best positioning to maximise the effectiveness, as well as practical considerations around space available in a room and access to power supply, cable routes, etc. Units should be positioned so that they do not interfere with the provision of care or provide an obstruction.
Units should always be positioned so that there is effective airflow into and out of the device. Vent panels on devices should not be blocked by furnishings and devices should be designed such that objects cannot be placed on top to cover vents. Patient comfort should also be considered with devices positioned such that they do not create uncomfortable draughts
Portable units can be a trip hazard in some locations and need to be positioned to ensure they or their cables do not pose a risk and do not impede access. Consideration should include risks for people who have visual impairments or restrictions on their mobility.
Consideration should be given to whether portable devices could be deliberately or accidentally moved or pushed over by patients or visitors. Device design should be stable and not easily toppled. In some settings it may be prudent to secure devices such that they cannot be moved.
8.5 Commissioning
Commissioning shall involve ‘acceptance testing’ according to local SOPs and include PAT testing to IEC 60601-1 (section 5.1.2). An audit of document compliance to the Low Voltage Directive is to be recorded. Where medical device classification is claimed, regulatory compliance with ISO 13485 Class 1 should be evidenced.
8.6 Verification and validation of performance
Manufacturers should evidence claims of engineering specifications (verification) and efficacy (bio-validation) (section 6.2). The air velocity and UVC irradiance in the reaction chamber should be characterised at an arbitrary point specified by the manufacturer (section 6.1).
8.7 Training
Staff in areas supported by UVC air scrubbing devices should receive training on operational and safety issues. A mechanism should be in place such that staff can notify estates management and/ or clinical engineering departments of suspected device malfunction. In an NHS context, such training might be included in staff mandatory training programmes.
8.8 Labelling
All UVC air scrubbing devices should be labelled to inform users of operating procedures and potential hazards. Labels should serve to make users aware of how to interact with UVC devices. Explicitly, these should include a hazard label to ISO 7010 ‘Non-ionising radiation’ and an indicative label ’Does not contain user-serviceable parts’.
9. Maintenance
Maintenance shall be conducted only by a designated competent person.
9.1 Cleaning
Cleaning of UVC lamps is not required during normal operation in most environments. However, if UVC lamps are used within environments that are particularly dirty, then cleaning might be necessary (section 8.2). Only cleaning products in line with the UVC lamp manufacturer’s recommendations should be used.
The outside surfaces of devices should be designed to be easily cleaned as part of standard cleaning regimes in the healthcare setting and should not have features which are prone to collecting dust and dirt. The device should be robust to cleaning materials.
9.2 Lamp replacement
After lamps have exceeded their active operational life, they shall be replaced. Old lamps shall be disposed of according to local SOPs (section 6.3.1).
9.3 Annual checks
All mobile UVC devices should undergo annual checks to verify their continuing performance. These checks should include, but are not limited to, the following:
- visual inspection of external and internal
- PAT test (5.1.2 Electrical safety)
- check alarms simulate failures
- check lamp run times and replace if necessary. (6.3.1 Effective life span).
- lean internals of the device.
- measure UVC irradiance level against manufacturer’s characteristic-specification (8.6 Verification of performance)
- replacement and safe disposal of any filters (8.2 Conventional HVAC filters)
- check air flow rate measurements at different speeds against manufacturer’s characteristic-specification (8.6 Verification of performance)
- check for UVC light spillage (4.1 Accidental exposure)
- check noise levels against manufacturer’s characteristic-specification (6.4 Noise considerations)
- apply annual check sticker.
10. Building Management System (BMS) module
The incorporation of a BMS module into UVC air scrubber devices is recommended to afford the assurance of effective operation and to support maintenance scheduling. Modules should be enabled with the BACNet* open protocol for interfacing with existing an BMS.
*BACnet is a communication protocol for building automation and control (BAC) networks using the ASHRAE, ANSI and ISO 16484-5 standards protocol.
Annex 1 – Historical reference to UVC effectiveness
Downes and Blunt demonstrate that sunlight prevents microbial growth:
- [H.1] Downes A, Blunt TP. Researches on the effect of light upon bacteria and other organisms. Proc R Soc Lond 1877; 26: 488-500.
Gates shows UV-spectral dependency with peak effectiveness around 265nm:
- [H.2] Gates FL. A study of the bactericidal action of ultra violet light: III. The absorption of ultra violet light by bacteria. J Gen Physiol 1930; 14(1): 31-42.
Wells proves the concept of infection via the airborne route and demonstrates the ability of UVGI to inactivate airborne microorganisms:
- [H.3] Wells WF. On air-borne infection: study II. Droplets and droplet nuclei. Am J Hyg 1934; 20: 611-8.
- [H.4] Wells WF, Fair MG. Viability of B. coli exposed to ultra-violet radiation in air. Science 1935; 82: 280-1.
Riley and Wells classic experiment which demonstrated that TB is airborne and that UVC reduces transmission:
- [H.5] Riley RL, Mills CC, O’Grady F, Sultan LU, Wittstadt F, et al. (1962) Infectiousness of air from a tuberculosis ward. Ultraviolet irradiation of infected air: comparative infectiousness of different patients. Am Rev Resp Dis 85: 511–525.
10.1 Reading list: recent peer reviewed papers demonstrating UVC effectiveness
Laboratory chamber studies demonstrating effectiveness of upper-room UV devices:
- [R.1] Ko G, First MW, Burge HA. The characterization of upper-room ultraviolet germicidal irradiation in inactivating airborne microorganisms. Environmental Health Perspectives, 2002; 110: 95–101. doi: 10.1289/ehp.0211095
- [R.2] McDevitt JJ,Milton DK,Rudnick SN,First MW. Inactivation of Poxviruses by upper-room UVC light in a simulated hospital room environment. PLoS One, 2008; 3: doi:10.1371/journal.pone.000318.
Efficacy of recirculating UVC units:
- [R.3] Corrêa TQ, et al. Efficiency of an air circulation decontamination device for microorganisms using ultraviolet radiation. Journal of Hospital Infection 2021; 115: 32–43. doi: 10.1016/j.jhin.2021.06.002
- [R.4] Snelling WJ, Afkhami A, Turkington HL, Carlisle C, Cosby SL, Hamilton JWJ, et al. Efficacy of single pass UVC air treatment for the inactivation of coronavirus, MS2 coliphage and Staphylococcus aureus bioaerosols. Journal of Aerosol Science 2022; 164: 106003. doi: 10.1016/j.jaerosci.2022.106003
- [R.5] Lee LD, Delclos G, Berkheiser ML, Barakat MT, Jensen PA. Evaluation of multiple fixed in-room air cleaners with ultraviolet germicidal irradiation, in high-occupancy areas of selected commercial indoor environments. Journal of Occupational and Environmental Hygiene 2002; 19(1): 67-77. doi: 1080/15459624.2021.1991581.
- [R.6] Qiao Y, Yang M, Marabella IA, McGee DAJ, Aboubakr H, Goyal S, et al. Greater than 3‑log reduction in viable coronavirus aerosol concentration in ducted ultraviolet‑C (UV−C) systems. Environmental Science and Technology 2021; 55(7): 4174-82. doi: 10.1021/acs.est.0c05763
Reduction in infection rates using various UVC approaches:
- [R.7] Menzies D, Popa J, Hanley JA, Rand T, Milton DK. Effect of ultraviolet germicidal lights installed in office ventilation systems on workers’ health and wellbeing: double-blind multiple crossover trial. Lancet 2003; 362(9398): 1785-91. doi: 10.1016/S0140-6736(03)14897-0.
- [R.8] Leach T, Scheir R. Ultraviolet germicidal irradiation (UVGI) in hospital HVAC decreases ventilator associated pneumonia. Ashrae Winter Conference, 2014
- [R.9] Escombe AR,Moore DAJ, Gilman RH, Navincopa M, Ticona E, Mitchell B, et al. Upper-room ultraviolet light and negative air ionization to prevent tuberculosis transmission. Plos Medicine 2009; 6: doi: 10.1371/journal.pmed.1000043.
Wider reading on UVC and air cleaning applications:
- [R.10] Wladyslaw Kowalski, Ultraviolet Germicidal Irradiation Handbook UVGI for Air and Surface Disinfection, 2009, Springer doi: 10.1007/978-3-642-01999-9
- [R.11] SAGE-EMG paper on air cleaning devices in the context of Covid-19
Annex 2 – Acknowledgements
NHS England would like to thank the Institution of Mechanical Engineers (IMechE), the Chartered Institution of Building Services Engineers (CIBSE), the Institute of Physics and Engineering in Medicine (IPEM), the NHS Innovation Agency (AHSN NW Coast) and the Office of The Chief Scientific Officer NHS England for their expertise in creating this document, and in particular our thanks go to the following colleagues who made expert contributions:
Authors
- Michael Ralph CEng FIMechE
- Professor Anthony Fisher MBE MD PhD FIET FInstP FIPEM FAHCS CEng CPhys CSci
- EUR ING Frank Mills BSc CEng FCIBSE FIMechE MASHRAE
- Paul Waldeck CEng MICE MIStructE MASHRAE MIMechE
- Professor Catherine Noakes OBE PhD CEng FREng FIMechE FIHEEM
- Mark Jackson TD VR MBA BEng(Hons) CEng FIMechE
- Stephen Clifford CEng BSc MCIBSE FIHEEM
Contributors
- Barry Paterson BSc CEng MIMechE
- Professor Fred Mendonça BSc MSc DIC ACGI
- Professor Chris Hopkins BSc MIET MIPEM FAHCS CEng
- John Cashmore MCGI FIHEEM FIIRSM RSP MCIBSE MIET CBuild E MCABE MIGEM CEng CMgr MCIM
Annex 3 – Glossary
- Absorption (light): Intake or retention of electromagnetic waves via conversion to heat, here: 254 nm wavelength radiation.
- Active operational life: A product’s operational life is the period for which a product is in use before it becomes obsolete, in terms of UVC lamps it is typically 70% of original efficacy.
- Aerosol generating procedure (AGP): An aerosol generating procedure refers to a health care treatment (eg dentistry/endoscopy) or event (cough/sneeze) which generates particulate matter referred to as droplets or aerosols.
- Air changes per hour: Air changes per hour (ACH) is the measurement at which air volume per hour is added to a room divided by the total volume of the room. It represents the number of complete air exchanges in one hour under perfect air circulation conditions. See also Equivalent air changes per hour.
- Air circulation: Mixing of the air from natural or mechanical ventilation sources inside an enclosure.
- Air circulation efficiency (%): A measure of the effectiveness of air circulation in a real enclosure with obstructions such as occupancy and furniture, compared with perfect mixing as quantified by ACH/eACH. CFD studies in hospital and high-street treatment rooms indicate that the air circulation efficiency can vary between 40% and 80% depending on the device placement and proximity of furniture, equipment and occupancy. Similar variance applies to AGP-clearance and therefore will affect fallow time.
- Age of air: Time (s/min/h) locally the air has been inside the enclosure/room at that location since entering from a fresh/clean/purified source (natural ventilation source, mechanical ventilation source or purification device). This is a useful measure of dead or recirculating air pockets in the enclosure volume.
- Apertures: Windows, doors and external vents connecting the enclosure to the outside atmosphere.
- Biofilm: Biofilms consist of a thin slime or dry layer (film) in which microorganisms (eg. bacterial or algae) are embedded. They form mainly in water systems, either on the surface of the water or on an interface with a solid phase. Inside the biofilms the embedded organisms are active and growing so that new microbes continuously are spread into the water. By this, for example, cooling systems and water reservoirs get steadily contaminated. Furthermore, on dying biofilms moulds and yeasts can settle down.
- BMS (Building Management System): A computer-based control system installed in buildings that controls and monitors the building’s mechanical and electrical equipment such as ventilation, lighting, power systems, fire systems and security systems.
- Building regulations: Building regulations set standards for the design and construction of buildings to ensure the safety and health for people in or about those buildings. They also include requirements to ensure that fuel and power is conserved, and facilities are provided for people, including those with disabilities, to access and move around inside buildings. Current standards require that healthcare buildings conform to NHS standards. For ventilation NHS HTM-03 applies.
- CFD (computational fluid dynamics): Computer-based fluid dynamics modelling providing a means to simulate air flow combined with convective/buoyant/conductive/radiative heat transfer, particulate transport (aerosols and droplets) and turbulence.
- Characteristic specification (Characteristic verification): A measurable property of the device that can employed routinely by the user to provide assurance of device operation to the verification model. See Verification.
- Clearance: The relative removal of a contaminant usually expressed as %. See Log reduction.
- Construction Design and Management (CDM) regulations: CDM regulations are a set of health and safety regulations that apply to every construction project in Great Britain.
- D90: Dose of UV to inactivate 90% of a microbial population. See k value.
- Decontamination: Decontamination describes the reduction of pathogenic microorganisms to a safe level for human use. Technically, this means reduction by a minimum of 1 log step, meaning 90%.
- Disinfectant: Disinfectants contain ingredients which either kill or inhibit the growth of microorganisms. Disinfectants require sufficient application time and must be used at sufficiently strong concentrations. Some well-known disinfectants are alcohols (eg. isopropanol), hydrogen peroxide (H2O2), ozone (O3) and tinctures containing iodine.
- Disinfection: The term disinfection is not clearly defined in a technical sense. Generally, for the purposes of this standard, it means a reduction of pathogenic microorganisms by a minimum of 3 log steps. Hence, the term ‘UVC disinfection’ describes the inactivation of at least 99.9% of a given pathogenic population with the aid of UVC technology.
- Dose: aka ‘Radiant Exposure’. The irradiance absorbed per unit time. Explicitly UV dose (μW·s.cm-2) = UV irradiance (μW.cm-2) × exposure time (s)
- Electromagnetic spectrum: The electromagnetic spectrum is the range of all frequencies of electromagnetic waves.
- Electromagnetic wave: An electromagnetic wave consists of an electrical and a magnetic field component. Unlike pressure waves, electromagnetic waves do not require a medium for propagation; their propagation speed depends on the medium, with propagation in a vacuum taking place at the speed of light. The best-known electromagnetic waves are probably those described colloquially as ‘light’.
- Emission: The sending out of electromagnetic waves.
- Emitter: The source of radiation is defined as an emitter.
- Epidemic: A localised, heavily massed occurrence of an infectious disease. See also Pandemic.
- Exposure time or dwell time: Length of time for which a microorganism is exposed to UVC irradiation (in the context of this standard).
- Equivalent air changes per hour, eACH: Equivalent air changes per hour, or eACH, is a measure of the ‘equivalent’ amount of air that is cleaned by a UVC device as a ventilation rate of new outside-air changes would achieve in one hour. See ACH. Note that this applies to decontamination and does not obviate the need for meeting minimum fresh air standards.
- Fallow time: Time (s/min/hr) allocated to a treatment room without occupancy to allow for clearance of the room after a contamination event (eg an AGP) to recover safe levels for occupancy.
- FDA: [Food and Drug Administration] – the FDA is the American federal agency responsible for food monitoring and drug licensing. It is subordinate to the Department of Health and Human Services.
- Fluence: The amount of irradiation (‘dose’) within an enclosed space to which the air being treated by UVC is subjected. Unit is mJ.cm-2.
- Fungicide: Chemical or biological agent for destroying fungal spores and moulds.
- Germicidal: Action destroying or deactivating a microorganism.
- Germicidal ultraviolet/germicidal ultraviolet irradiation: Referred to commonly as GUVC and UVC. Both are one and the same in that they refer to ultraviolet C spectrum light that is germicidal.
- HACCP: [Hazard Analysis and Critical Control Points] – a preventive system intended to ensure food, medicines and safety critical products safely from manufacture to the consumer.
- Hazard assessment: A hazard assessment is a thorough check of the occupational environment. The purpose of a hazard assessment is to identify potential risks and hazards in the area, as well as to identify appropriate safety measures to be used to mitigate, eliminate or control the identified hazards.
- HAZOP: [Hazard Analysis and Operational study] – a systematic way to identify hazards in a work process.
- IAQ (Indoor Air Quality): A generic term used for air quality in enclosed spaces, usually referring to the combination of harmful gases (eg. CO2 and CO levels measured in parts-per-million, ppm), temperature (for thermal comfort), total volatile organic content (TVOCs measured in parts-per-billion, ppb), relative humidity (%) and particulate matter size (respiratory irritants/hazards) measured in microns-diameter, eg. PM2.5, PM10.
- Inactivation: Prevention of microbial replication.
- Infection: The process by which pathogens penetrate the body of an organism and multiply therein. Depending on the transmission route, we distinguish between contact infections and airborne infections.
- Infectiousness: Measure for describing the ability of a pathogen to cause actual infection in a host after transmission occurs.
- Intensity: In physics, ‘intensity’ describes energy density with respect to area.
- Ionising radiation: Ionising describes the type of radiation capable of permanently removing electrons from atoms or molecules. Note: UVC radiation has no ionising power (See also Technology – generating UVC rays).
- IP rating: [Ingress Protection] – types of protection that are classified according to IEC standard 60529. The letters IP are followed by two digits, the first indicating the degree of protection afforded against the ingress of solid bodies, and the second describing the degree of protection against the ingress of water.
- k value: Inactivation rate constant (susceptibility rate) k = (-ln(1-0.9))/D90. Units cm2.mJ-1.
- Lethal dose: Lethal dose (LD) is the term referring to the dose of a toxin or radiation which is deadly or inactivates an organism (this term includes microorganisms).
- LD 90: LD 90 is the dose which eliminates or inactivates on average 90% of an organism’s population.
- Lethality: Lethality describes the ratio of deaths/eliminations/inactivations to survivals after a dose of radiation, infection, or illness viz the ‘mortality rate’.
- Living organism: In biology, life forms capable of metabolic processes, replication and evolutionary development (all three criteria must be fulfilled) are known as living organisms.
- Log: [common logarithm] – although the term ‘log’ is the usual abbreviation for base-10 logarithms, the mathematically correct term here is log10. We speak here of decadic logarithms.
- Log reduction: The reduction of a contaminant can be quantified in log stages. A Log reduction of ‘x number’ therefore means a reduction by ‘x number Log’ stages starting from a given population. The reduction by 1 log stage means a reduction of 90%, since only 10% have survived from the original population. See Clearance.
- Log stage (a.k.a. Log step): A log stage or log step describes the reductio n of a population by a (further) power of ten: in other words, 1 log stage = 90%, 2 log stages = 99%, 3 log stages = 99.9%, etc. See Log reduction.
- Melanoma: Also known as black-mole cancer – a melanoma is a malignant tumour appearing as an asymmetrically growing, discoloured change in the skin.
- Microorganism (microbe): A microorganism is an organic structure so small that they can generally only be seen with the aid of a microscope and include viruses, bacteria and fungi. Such structures are usually single-celled, although they are occasionally multi-celled.
- Monochromatic: Describes radiation of a precisely defined wavelength, as, for example, emitted by a laser.
- Mutation: The changing of the structure of a gene, resulting in a variant form that may be transmitted to subsequent generations.
- Nosocomial infection: An infection contracted in a hospital or care institution.
- Optical radiation: The electromagnetic wavelength range between 100 nm and 1 mm is referred to as optical radiation. This includes ultraviolet radiation (UV), the visible light spectrum (VIS) and infrared radiation (IR).
- Organism: An organism is an individual life form. See Living organism.
- Ozone: Represented as O3. Ozone is a gas with strong oxidation properties that is toxic in low concentrations. Ozone can result from the oxidation of O2 irradiated by far UVC.
- PAT (portable appliance testing): Requirement of the Low Voltage Directive to demonstrate general electrical safety.
- Pandemic: A pandemic is an infectious disease of temporarily exceptionally high prevalence occurring across national borders. See also Epidemic.
- Pandemic resilience: Pandemic resilience is the ability to withstand, protect and recover quickly from any pandemic by ensuring infrastructure and buildings are equipped with the necessary safeguards to combat, eliminate or control pathogenic hazards that are so prevalent as to be classified as a pandemic or endemic hazard.
- Pathogen: Pathogens are microorganisms capable of causing disease or illness in living creatures.
- Prevention: The taking of precautionary measures to stop undesirable occurrences.
- Radiometer: A radiometer serves to measure electromagnetic power These devices are generally based on photodiodes which convert the incoming radiation into a proportional electrical signal.
- Radiometry: Radiometry is the science of radiation measurement.
- Reflection: The (partial) return of electromagnetic waves at an interface. Reflection is the opposite of absorption. UVC air cleaners will be fitted with highly reflective materials within the air passageways in order to reflect and thereby amplify the amount of UVC in the air.
- Residence time: The average time taken by the air or airborne particles to pass through the UVC fluence zone. Unit seconds (s).
- Sanitisation: The process of reducing microbiological contamination. See Clearance and log reduction.
- Sensitivity: Here: responsiveness or susceptibility to UVC radiation. See k value.
- SOP: (Standard operating procedure) A set of step-by-step instructions compiled by an organization to help workers carry out routine operations.
- Sound level: dB3m: The acoustic power represented by dB measured at 3 m from the source.
- Target: A person, organism or thing that receives or is infected by an intervention.
- Toxic: The effect of a toxin is described as toxic. ‘toxic’ can also be defined as meaning ‘poisonous’.
- Toxicity: The degree to which a toxin is toxic or poisonous.
- Toxin: A toxin is a biogenic substance capable of damaging an organism by disrupting its physiological metabolic processes. The scientific discipline investigating toxins is called toxicology.
- UV spectra: The UV spectrum is commonly sub-divided into four regions:
- Far UV or vacuum UV: [100…200] nm
- UVC: [200…280] nm (NB germicidal UV)
- UVB: [280…315] nm
- UVA or near UV: [315…400] nm
- Validation (bio-validation): The process to provide assurance that the device is effective as claimed by the manufacturer. For the purposes of this standard, assurance that sanitisation is achieved as claimed.
- Verification: The process to provide assurance that the device performs to the manufacturer’s specification. For the purposes of this standard, assurance that air flow and UVC dose are as claimed.
- Viruses: Viruses are particles or information carriers dependent for survival and replication upon the metabolism of a host cell since they themselves have no cytoplasm and are incapable of metabolism. Viruses are thus, de facto, not living organisms.
The National Estates and Facilities team at NHS England is responsible for producing Standards and Guidance for the NHS estate and ensuring that the information and guidance they contain remains up-to-date and relevant for users.
NHS Estates Technical Bulletins (NETBs) enable updated guidance to be passed to local systems, ensuring we maintain our focus on patient safety. NETBs contain technical guidance and standards which systems and organisations are required to consider and implement, where applicable. Boards are responsible for their assessment and application to their organisations.
Date of issue: 9 May 2023
NHS Estates reference: NETB 2023/01B
Publication reference: PR1324_i

