The latest on NHS England’s five-year strategy for specialised services
My latest blog is coming from day one of the Health and Care Innovation Expo 2014 event in Manchester. Members of the national specialised commissioning team will be present at the main NHS England exhibition stand between 9am and 2pm both today and tomorrow, so please do come along and talk to us and find out more about the development of our five-year strategy for specialised services.
One of the key activities for specialised commissioning at EXPO is a round table meeting tomorrow with the provider organisations which deliver specialised services care. This is the second of two such meetings held with providers, aimed at developing a Provider Reference Group, which will be the main route for engagement with trusts during development of the five-year strategy.
NHS England stated in its planning guidance that it will be ‘looking to reduce significantly the number of centres providing NHS specialised services; require standards of care to be applied consistently across England, and maximise synergy from research and learning’.
A critical element of the five-year strategy for specialised services will be achieving a balance between improved outcomes of care and ensuring that patients remain able to access high quality services, regardless of where they live. Working with providers therefore is critical to this aspect of the strategy work, as we will be looking for innovative ways of working which can deliver care closer to patients’ homes.
This week, NHS England published details of how it will aim to fulfil the 51 commitments as set out in the first UK Strategy for Rare Diseases.
The Strategy was published by the Department of Health in November last year, and all four devolved nations were charged with producing a Statement of Intent, setting out their plans for implementation.
NHS England published its Statement of Intent on 26 February to coincide with a parliamentary reception, attended by myself and Earl Howe, and hosted by Rare Disease UK.
We worked closely with Rare Disease UK in the production of the document and look forward to collaborating with them in the future development of the five-year strategy, of which our approach to rare diseases will play a major part.
I particularly welcome the appointment of the new Department of Health Rare Disease lead, the Deputy Chief Medical Officer, David Walker. David’s appointment will enable us to maintain critical links with Government as we work through implementation of the strategy.
We are currently in the engagement phase of our five-year strategy development. We held a series of ten very successful area team events, attended by hundreds of patient representatives, clinicians, commissioners and other key stakeholders around the country.
There were some very lively discussions about a wide range of topics, many of which were included in our initial engagement document.
A number of the area teams recorded their discussions as visuals. An example of these graphic records from the East Anglia event can be found below. Click on the image to see all records and notes from the engagements events.
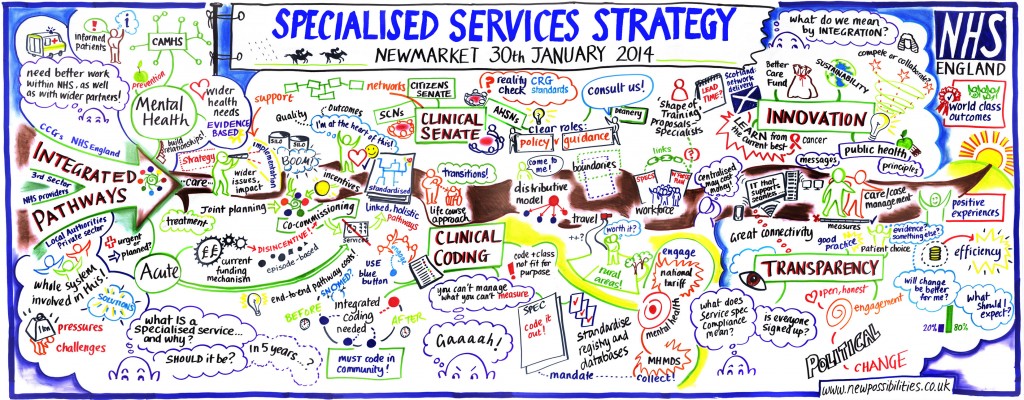
Thanks to all those who both organised, and attended these events. The feedback received will be used to inform our final strategy.
Today sees the publication of yet another key document which will be used in the development of our strategy.
The ‘Vision for Radiotherapy’ (Gateway reference number 01261) is a joint publication, produced by ourselves and Cancer Research UK. It sets out our shared aspirations for the future of the radiotherapy cancer care across England.
Thanks to our collaborative approach to working with industry, we now know what cutting edge treatments we can expect to see on the market within the next 5 to 10 years, and can take these into account when developing our future strategy for radiotherapy services.
There are a number of challenges in making this vision a reality, not least funding, which is why the findings of this report will be fed into the wider five-year planning. The ‘vision’ document will form a key foundation for this work.
We also held a very successful industry-led event as part of the formation of an Industry Reference Group. This group will act as the main route for engagement with industry during the development of the strategy and this initial meeting attracted more than 60 representatives from different organisations within this sector.
The establishment of this group is a key part of our stakeholder engagement activity. Industry colleagues provide us with expertise and knowledge about innovations on the horizon which will enable us to ensure that our final strategy takes account of those cutting edge developments which will help to improve patient outcomes and the overall patient experience of specialised services care.
Feedback from this event will be written up and published on the NHS England website shortly.
The next phase of defining the future strategy is to consider the issues for each of the clinical services we coordinate through our 75 Clinical Reference Groups. Carrying on with the engagement theme, we will shortly be completing a series of five Programme of Care events at which we will be discussing how the themes identified so far can be developed within the clinical services.
These events will include representatives of our Clinical Reference Groups, commissioners, as well as invitees from clinical commissioning groups, and local authorities and their Health and Wellbeing Boards.
To get involved in the development of the five-year strategy for specialised services, keep checking our web pages for details of events and publications.

One comment
Dear Sir,
I´m writing to you as the leader of the Stockholm County Council HTA-program. We are in the process of searching evidence for Middle Ear Implants and have found your CC Policy Statement NHSCB/D09/PS/a. We understand that it is an interim recommendation awaiting a formal NICE Technology Appraisal Guideline.
I wonder if that is correct and when you expecthe definite appraisal?
Sincerely
Elisabeth Persson
Medical advicer, Assoc.prof.
Docent, Medicinsk rådgivare
Ansvarig Metodrådet SLL-Gotland
Avd. Stöd för Evidensbaserad Medicin
Hälso- och sjukvårdsförvaltningen
Stockholms Läns Landsting
tfn 073 966 19 19