Foreword: Best practice timed diagnostic pathways
Best practice timed pathways support the ongoing improvement effort to shorten diagnosis pathways, reduce variation, improve experience of care, and meet the Faster Diagnosis Standard (FDS).
The 28-day Faster Diagnosis Standard remains the single standard by which the speed of diagnosis of cancer will be measured under Cancer Waiting Times. However, some types of cancer are particularly aggressive, and particular effort should be made to diagnose them even faster than the headline target. Many hepato-pancreatic biliary (HPB) cancers fall into this category, and the pathways in this document, which set out the sequence of events that would be required to complete the diagnostic process wherever possible within 21 days are reflective of that.
This HPB pathway is part of a series, which includes the oesophago-gastric pathway and completes the pathways that cover upper gastrointestinal cancers, published since April 2018. From previous pathways implemented by Cancer Alliances, Implementation Guidance was shared in June 2021, identifying areas that are key to success, such as setting up with clinical and operational engagement, auditing pathways, allocating project management resources, ensuring leadership, analysing data, and sharing successes.
This guidance complements existing resources such as NICE Guidelines (including NG12) and British Society of Gastroenterology guidelines and should therefore be read alongside such guidance. While the guidance stipulates recommended clinical actions and timings to support pathway design, we recognise that responsibility for clinical decision making remains with local clinical teams with the knowledge and expertise to make appropriate decisions and policies.
The pathway in this document was developed by a multi-disciplinary consensus group with clinical leaders from local and specialist services across England, and expert advice from Cancer Alliances, charities, and people with lived experience.
For any questions about this document please email england.cancerpolicy@nhs.net.
Dame Cally Palmer, National Cancer Director, NHS England
Professor Peter Johnson, National Clinical Director for Cancer, NHS England
Professor Paula Ghaneh, Chair of the HPB Task and Finish Group
The Faster Diagnosis Standard
We committed in the NHS Long Term Plan to provide a faster diagnosis for people through the introduction of the Faster Diagnosis Standard (FDS). This standard will ensure people are told they have cancer, or that cancer is excluded, within a maximum of 28 days from referral. The new standard is intended to:
- reduce the time between referral and diagnosis of cancer
- reduce anxiety for the cohort of people who will be diagnosed with cancer or receive an ‘all clear’
- reduce unwarranted variation in England by understanding how long it is taking people to receive a diagnosis or ‘all clear’ for cancer.
- represent a significant improvement on the current two-week wait to first appointment target, and a more person-centred performance standard.
FDS performance data, including a breakdown by suspected cancer pathway, has been published since June 2021, and faster, more streamlined pathways will be a priority.
As the key system-wide organisations for cancer services, Cancer Alliances will need to work across the local system to ensure that implementation is prioritised by senior stakeholders, clinical leaders, and operational colleagues, and that capacity is prioritised to enable the standard to be delivered.
The FDS has been formally performance managed since October 2021 activity, in line with cancer services recovery, with an initial threshold of 75%, rising to 80% in 2025/26. Cancer Alliances will need to ensure that they have plans to meet the threshold, which will need to be increased in subsequent years if we are to contribute to achieving the early diagnosis ambitions in the NHS Long Term Plan.
The case for change
Upper GI and HPB cancers
- Suspicion of Upper GI (HPB and oesophago-gastric [OG]) cancer is the sixth most common suspected urgent referral type in England, representing 7.5% of all urgent suspected referrals in 2020.
- Between 2018 and 2019, 70% of patients diagnosed with Upper GI cancer (HPB and OG) commenced treatment within 62 days of referral. This varied by Cancer Alliance from 60% to 86%.
- Between 2020 and 2021, 64% of patients diagnosed with Upper GI cancer (HPB and OG) commenced treatment within 62 days of referral. This varied by Cancer Alliance from 49% to 79%.
- HPB cancers affect more than 18,000 people each year, with more than 16,000 people dying each year.
Primary liver cancers
- In 2018, the median wait between referral and start of treatment was 56 days. This varied by Cancer Alliance with a range of 32 to 97 days, demonstrating the potential scope for improvement by standardising the pathway and reducing variation.
- For cancer patients in England diagnosed between 2015 and 2019, five-year age-standardised net survival was only 13.8% for Liver cancer.
Pancreatic cancers
- Pancreatic cancer is the quickest killing cancer, with one in four people dying within a month and three in four within one year. Pancreatic cancer has rapid disease progression and people experience nutritional, physical, and psychological deterioration rapidly following a diagnosis. Therefore, delays to diagnosis and treatment can lead to people quickly becoming inoperable or too unwell to tolerate treatment.
- Pancreatic ductal adenocarcinoma has the lowest early stage diagnosis of all common cancers, with six times higher one-year survival at early stage compared to people diagnosed at stage four.
- Pancreatic cancer is the deadliest common cancer, with 8% one-year survival and 7.8% five-year survival in England, for those diagnosed between 2015 and 2019. This represents over 9,000 cases and over 8,000 deaths every year in England. There is also variation in pancreatic cancer survival within England, with one–year survival ranging from 22.5% to 33.3% and five year survival ranging from 5.0% to 11.3% across Cancer Alliances, for those diagnosed between 2015 and 2019.
- A streamlined and more efficient pathway will improve avoidable delays and should reduce the variation currently seen.
Cholangiocarcinoma
- Cholangiocarcinoma has a poor prognosis with 80 to 90% of patients presenting with inoperable disease and 50% presenting in emergency departments with symptomatic disease.
- The incidence of intrahepatic cholangiocarcinoma is increasing and the incidence of biliary tract cancer as a whole is at least equivalent to hepatocellular cancer.
- 50% of patients receive no active therapy despite demonstrated benefit of multiple interventions including NICE recommendations.
Hepatocellular carcinoma
- Hepatocellular carcinoma (HCC) is closely linked to liver cirrhosis and chronic viral hepatitis, more than 80% of patients diagnosed with HCC also have underlying liver disease.
- The incidence of HCC is increasing throughout the UK, and one year survival remains below 50%.
- Liver disease can be asymptomatic and most people with HCC are still diagnosed at an advanced stage via emergency presentation or GP referral.
- Early detection of both liver disease and hepatocellular carcinoma will increase the proportion of patients diagnosed at an early stage when potentially curative treatment could be offered.
- Patients with chronic liver disease should be offered regular tests aimed at early detection of HCC, based on British Society of Gastroenterology guidelines for the management of hepatocellular carcinoma in adults and NHS England’s Hepatocellular carcinoma surveillance: minimum standards.
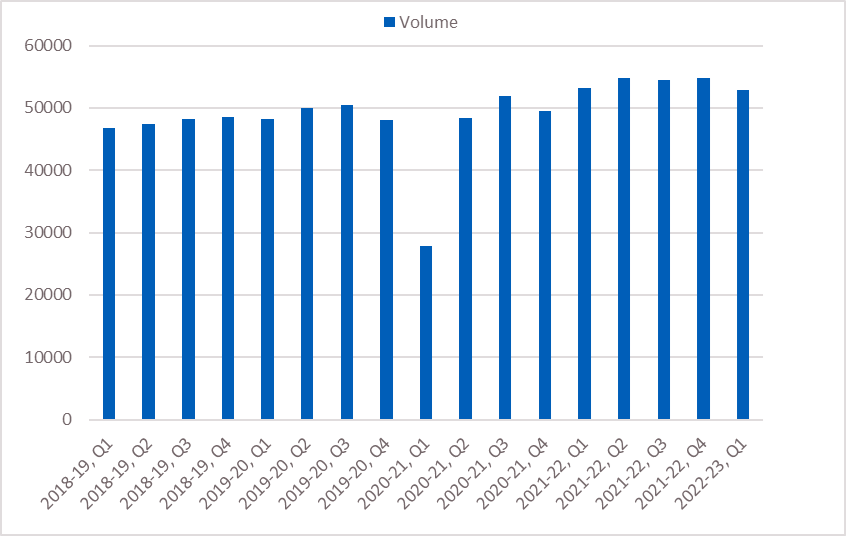
Figure 1: Upper GI (HPB and OG) cancers referred for urgent suspected cancer (two-week wait standard) by volume, 2018/19 to 2022/23 (up to quarter one)*
*Two-week wait data for upper GI is included as a proxy for the FDS to demonstrate pre-COVID volumes as a baseline, because official FDS data is not available before April 2021.
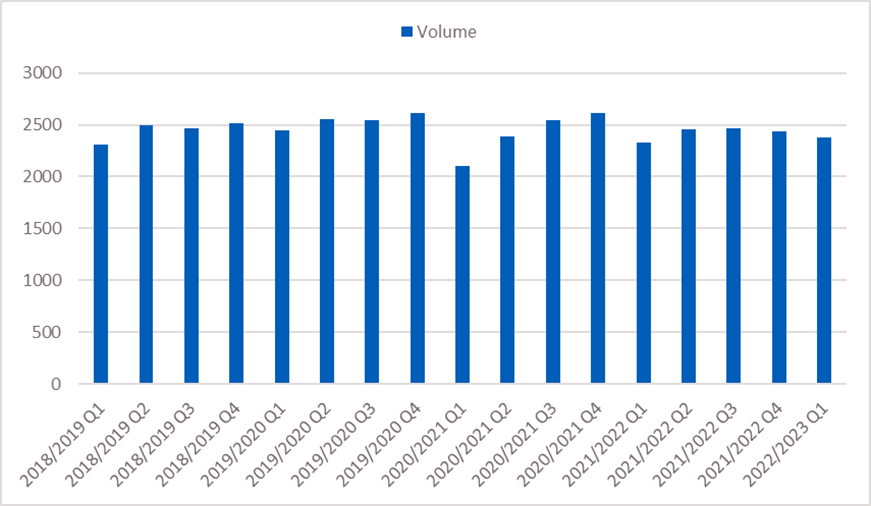
Figure 2: Upper GI (HPB and OG) cancers referred for urgent suspected cancer and commencing treatment (62-day standard) by volume and performance, 2018/19 to 2022/23 (up to quarter one)
| “The patient pathway must consider a combination of imaging tests and biopsy, if appropriate, and enable onward referral. Initial diagnostics can be undertaken at local units and the more invasive staging investigations may require specialist decision and intervention.
The key features of these pathways are to implement rapid straight to test provision for multiphase CT or ultrasound, with hot reporting and assessment of results. This should be integrated with timely booking and reporting of staging investigations to support treatment commencing in a timely manner. The proposed changes are simple but do require better planning, capacity, and administration. Their implementation is anticipated to reduce waiting times for critical investigations and enable prompt starts for treatment for those diagnosed with cancer.”Paula Ghaneh, on behalf of the HPB Task and Finish Group, NHS Cancer Programme |
Benefits of pathway change
For patients and carers
- reduced anxiety and uncertainty of a possible cancer diagnosis, with less time between referral and receiving the outcome of diagnostic tests
- improved patient experience from fewer visits to the hospital, particularly to specialist centres if possible, and avoiding emergency admission
- potential for earlier recognition and initiation of pre-optimisation for treatment that could reduce complications and adverse outcomes
For systems
- reduced demand in outpatient clinics with increased straight to test provision and use of pathway navigators
- improved quality, safety, and effectiveness of care with reduced variation and improvement in outcomes
- allow resources to be targeted at patients with cancer by removing non-cancer patients earlier in the pathway
Experience of care
- patients and carers know they are urgently referred for investigation of suspected cancer and should expect a rapid diagnosis
- ensure that patients and carers’ ability to attend appointments is taken into account and additional support is offered, where necessary
- patients are communicated with clearly, understand the information provided, and are given additional support, such as access to a clinical nurse specialist (CNS) or navigator, psychological support, buddy system, where necessary
For clinicians
- using a nationally agreed and clinically endorsed pathway to support quality improvement and reconfiguration of pancreatic, liver, bile duct and gall bladder cancer diagnostic services
- using predetermined diagnostic algorithms and standards of care to streamline clinical decision-making and reduce delays for MDT discussion
- improved ability to meet increasing demand and ensure best use of the highly skilled workforce
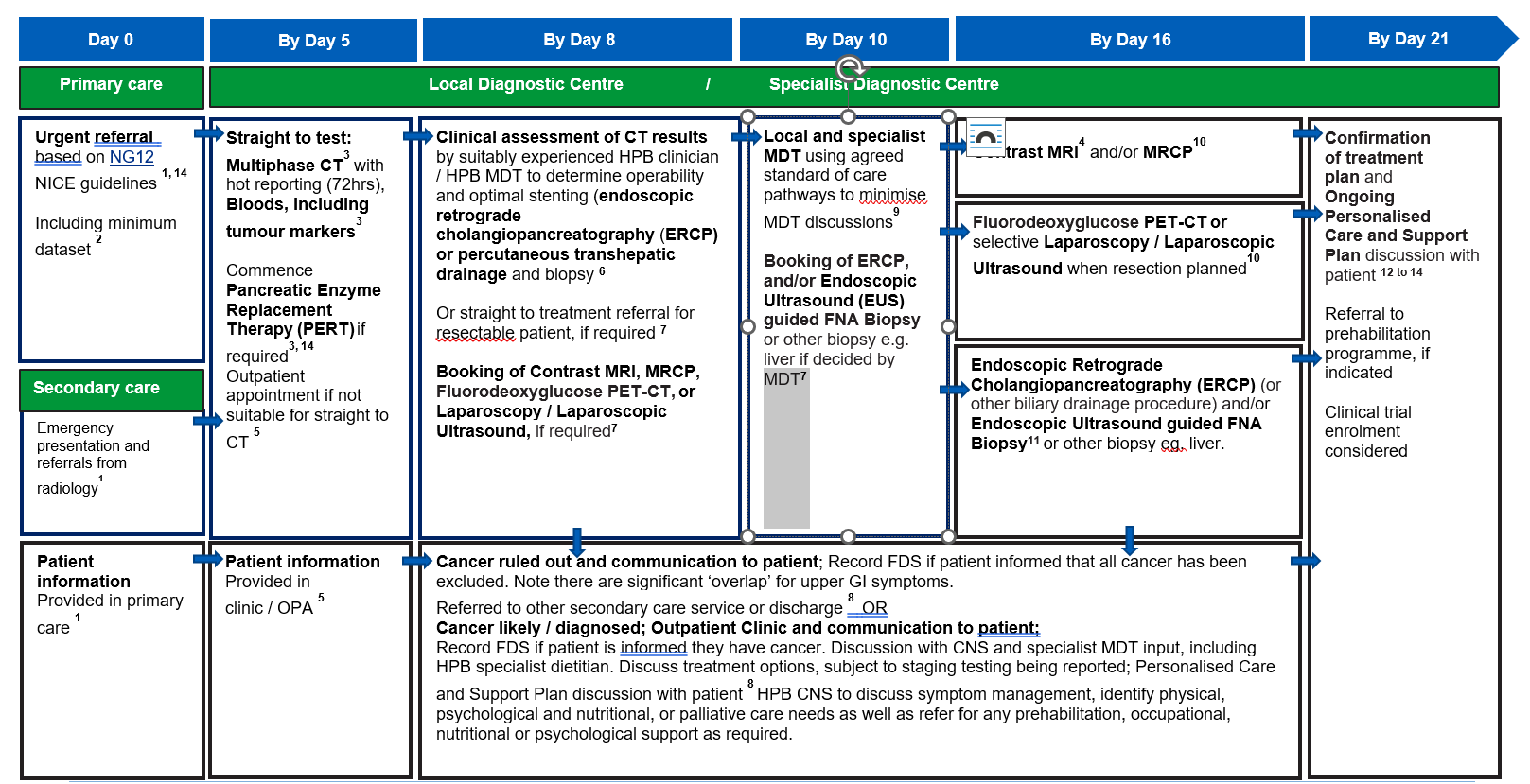
Jaundice, pancreatic, extrahepatic cholangio, gall bladder
See detailed information section for further information.
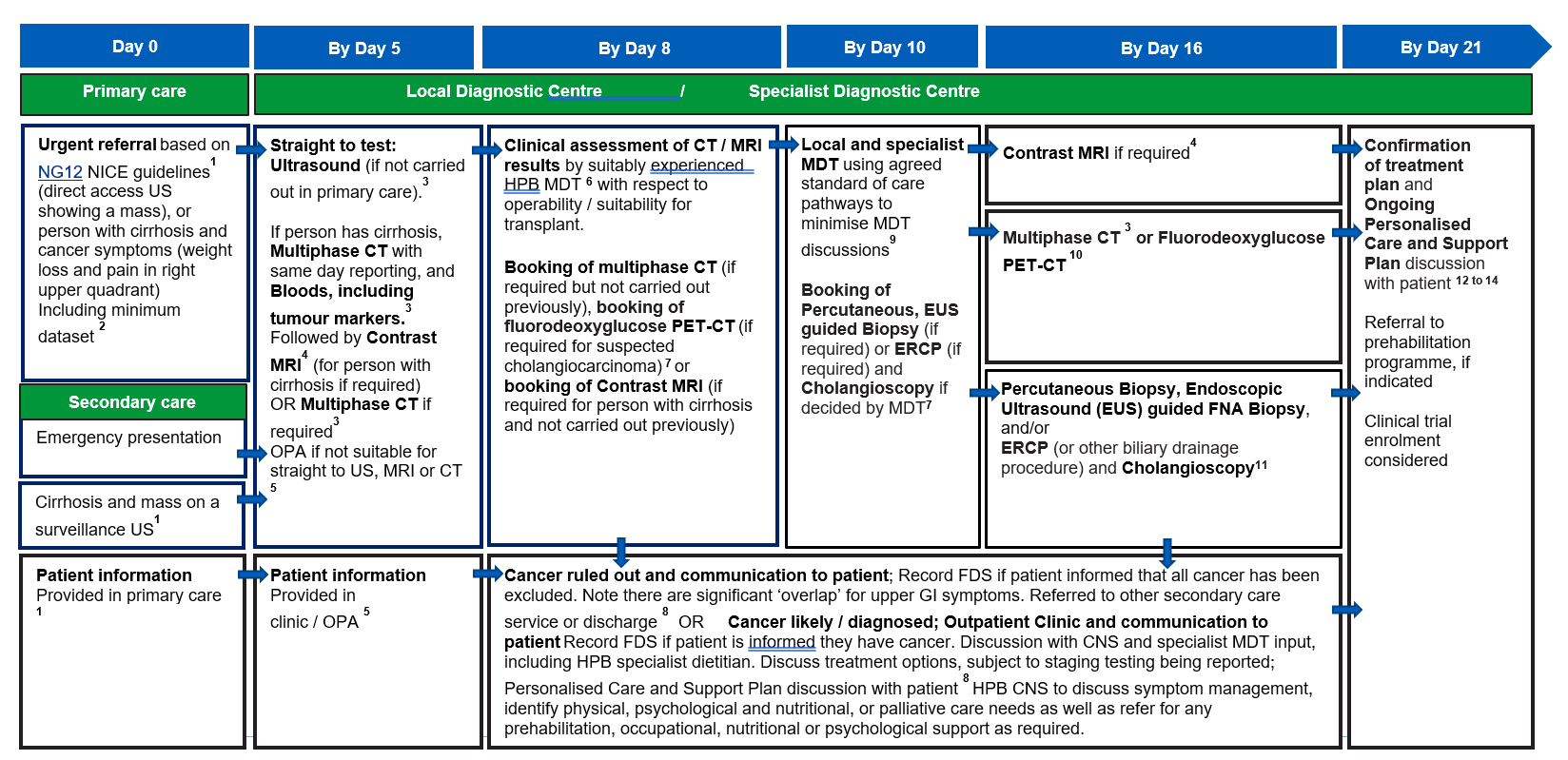
Hepatocellular carcinoma and intrahepatic cholangiocarcinoma
Some patients with pancreatic cancer may present within this pathway with overlapping symptoms. These patients may have scans carried out in this pathway and cross over to the jaundice, pancreatic, extrahepatic cholangiocarcinoma and gall bladder pathway to complete diagnosis. See detailed information section for further information.
See detailed information section for further information.
Detailed information
- An urgent referral pathway should be used for patients who meet NG12 criteria for suspected cancer pathway referral.
It is also recommended by the HPB Task and Finish Group membership that specialists should raise awareness with primary and community care, of the following symptoms associated with pancreatic cancer, in addition to NG12 symptoms, to reduce emergency admissions and support timely diagnosis:
- appetite loss, dark urine, steathorrhoea, pale stools, thirst, reflux, dyspepsia, bowel change
- people under 60, who present with symptoms consistent with NG12
- people presenting with jaundice at any age
- people aged over 50 with new onset diabetes with atypical presentation (e.g. low BMI, low weight, weight loss) or with existing diabetes and unexplained sudden loss of glucose control
It is also recommended that specialists raise awareness with primary and community care regarding early detection of liver disease and testing for viral hepatitis in at risk groups.
If someone diagnosed with pancreatic cancer also has a family history of pancreatic cancer, consider referring family members to the EUROPAC trial for at risk surveillance.
For suspected liver cancer, people presenting with jaundice will be directed through the jaundice, pancreatic, extrahepatic cholangiocarcinoma and gall bladder pathway. People with cirrhosis and cancer symptoms, such as weight loss and pain in right upper quadrant, should be referred urgently on to the hepatocellular carcinoma and intrahepatic cholangio carcinoma pathway.
People with cirrhosis who have a liver lesion suspicious for HCC detected on surveillance imaging should proceed to urgent investigation and should be managed according to British Society of Gastroenterology / HCC UK guidelines for the diagnosis and management of HCC. Early referral to a specialist HCC MDT is strongly recommended.
For suspected cholangiocarcinoma pathway, patients should be managed according to the British Society of Gastroenterology guidelines for diagnosis and management of cholangiocarcinoma.
While it is important to identify in the referral if there are specific concerns about a particular cancer site (i.e. pancreas, or liver) to support secondary care to ensure comprehensive coverage of diagnostic tests (i.e. if CT needs to consider pancreatic and OG cancer), it is equally important to consider the general holistic needs of the patient.
There is significant ‘overlap’ for upper GI symptoms, so for anyone aged 55 or over with weight loss and upper abdominal pain, vomiting, nausea, reflux, or dyspepsia, the HPB Task and Finish Group recommends the patient should have an OGD and CT of the pancreas, when referred on a suspected cancer pathway to Upper GI teams.
For people who present with abdominal pain and weight loss, consider assessing for both pancreatic and colorectal cancer.
Where a GP refers the patient for a direct access test and the CT or ultrasound is abnormal and suspicious of cancer, patients should be followed up directly by secondary care, without the need for an urgent referral from their GP. The patient should then join the pathway after the first diagnostic test (labelled on this pathway diagram as ‘straight to test’).
Similarly, where a patient receives initial tests in A&E and they are still suspicious of cancer, they should be upgraded on to the pathway after the relevant test(s) are taken under the care of A&E. They should not be referred back to their GP. The National Cancer Waiting Times Monitoring Dataset Guidance v12.0 sets out consultant upgrade rules, including escalation of direct access tests and from non-GP scenarios such as A&E and acute settings.
Primary care should provide information to the patient, including information about FDS and the urgent suspected cancer pathway, expected timelines including that the patient should be available within the next 14 days for appointments and tests.
As part of recovery of services and effective referral management, Cancer Alliances are encouraged to use this guidance and other policies to support work locally, with commissioners, to provide educational opportunities for GP practices on cancer symptoms, urgent referral processes and the locally agreed minimum dataset. This should include hard to reach and outlier GP practices.
- A minimum dataset to accompany the referral and facilitate straight to clinic and immediate diagnostics, to be agreed locally, should include:
- description of referral reason in line with NG12 guidelines
- patient demographics
- estimated Glomerular Filtration Rate (eGFR) if already provided in last three months
- full blood count
- urea and electrolytes (U&E)
- renal function including creatinine
- anticoagulant status
- performance status
- co-morbidities including diabetes status
- dementia
- mental health conditions, such as claustrophobia
- smoking history
- Body Mass Index
- prescribed medication
- allergies
- family history of cancer
- functional / frailty score (E.g Rockwood)
- presence of metal implants or pacemakers
- need for an interpreter
- mental capacity to consent.
Referrals cannot be rejected by secondary care if minimum dataset items are not included – further information is available in the National Cancer Waiting Times Monitoring Dataset Guidance v12.0.
- CT further information: Ideally this would be triple phase pancreas protocol CT of the abdomen and pelvis for patients with suspected pancreatic cancer, and multiphase examination for patients with suspected cancer of the liver or gall bladder.
CT of the chest should be added for all patients if there is a history of weight loss or painless jaundice. The abdominal CT should always be performed prior to any endoscopic procedure, which can in some instances lead to complications that can mask the site and extent of the tumour. NICE’s NG85 guidelines should be followed where appropriate.
For suspected pancreatic cancer, the specialist MDT review should ideally include a radiology reporting template, such as PACT-UK template, to ensure consistency in reporting of findings. There is significant variation in radiological reporting across England, including reporting of vascular involvement, with 100% concordance in only 30% of cases.
Bloods include checking for FBC, U&E, LFT, clotting, if not provided in primary care, HbA1c, AFP tumour marker (for suspected HCC) and CA 19-9 tumour marker (for suspected pancreatic cancer, cholangiocarcinoma and gallbladder cancer).
Pancreatic Enzyme Replacement Therapy (PERT) is indicated for patients with suspected pancreatic cancer. Only 50% of people with pancreatic cancer receive PERT, despite being included in the NG85 pancreatic cancer guidelines. Therefore, Cancer Alliances and local stakeholders should take action to improve local healthcare professional awareness of PERT.
An online healthcare professional course about PERT is available, endorsed by the British Dietetic Association and Pancreatic Society of Great Britain and Ireland. Prescribing PERT for people with inoperable pancreatic cancer is included in the NICE quality standards for pancreatic cancer.
If symptoms consistent with pancreatic exocrine insufficiency show prior to diagnosis, PERT should be immediately prescribed so that the patient doesn’t deteriorate in advance of any cancer treatment, should cancer be subsequently diagnosed. This can be prescribed in primary care if agreed locally.
There should also be consideration of wider nutritional requirements, including oral nutritional support, micronutrient replacement, medication for high blood glucose levels and consideration for laxatives, antiemetics and proton pump inhibitors (PPIs) if required.
An endoscopic ultrasound should ideally be carried out by an expert ultrasound operator, in clinic or externally provided, and to a locally agreed minimum reporting dataset and algorithm. If an ultrasound is not high quality then the next step should be rapid referral for an expert ultrasound scan.
- Standard imaging protocols should be applied for all MRI and these should comply with Royal College of Radiologists’ recommendations for pancreas and liver. Ring-fenced general cancer MRI slots should be considered to ensure that capacity is available to deliver expected imaging within seven calendar days of assessment of CT results. MRI should be carried out to minimal standards, to avoid the requirement for repeat procedures.
For patients with persistent symptoms, raised CA 19-9 and normal scan, consider using the local non-specific symptoms pathway where there is no other obvious cause for the symptoms and finding. These patients can have a significant incidence of malignancy in other sites.
- Some patients may not be suitable for straight to test, or if complex may require a face-to-face outpatient appointment. An outpatient appointment (OPA) should be provided for this cohort of patients (those medically unfit for straight to one-stop or that require a face-to-face appointment). The recommended first line investigation should be performed and reported within four days of OPA so that this cohort can progress on the pathway in the same timeframes.
If a suspicious lesion is identified, patients should have access to a CNS with expertise in HPB cancer for support (from this stage onwards). Patients who attend OPA should have same-day tests to reduce repeat visits and improve patient experience. If this is not possible, tests should be on the next day (i.e. within 24 hours). Consider ring-fencing urgent cancer slots in advance and releasing them if no longer required.
Patients and carers should be asked what information they require about the pathway, provided with standard leaflets about investigations when sending confirmation of appointment, confirmation of next step(s) and anything the patient needs to do to prepare for the day (e.g. can they eat and drink before), and whether they have any disabilities or language barriers.
Preferences for the amount of information and when it is provided will vary, and therefore it will help to provide pathway navigator telephone contact details who will be able to provide support throughout the pathway and outside of clinic times. They should also provide signposting to charities and support services, provide information about caregivers attending appointments, and offer follow-up if patients do not receive confirmation of an appointment in expected timescales.
Where possible, continuity of caseworker/navigator should be provided to enable familiar contact and to build trust. Patients should also be informed ahead of time that it is likely they will receive a procedure and/or diagnostic test on the same day at the first face-to-face appointment.
The clinical triage consultation or first OPA is also an opportunity to collect minimum dataset items from the patient, if not provided in the primary care referral. It is also an opportunity to consider psychosocial and mental health factors and vulnerabilities that could interfere with engagement of patients in the pathway and ensure that proper mitigation plans are in place.
The patient navigators should be able to signpost and use third sector organisations’ training, patient information and additional support services, such as a Specialist Pancreatic Cancer Nurse Support Line, patient information, online patient support sessions, and cholangiocarcinoma patient resources and this should be included as part of their role description.
- For suspected pancreatic cancer, clinical assessment can be undertaken by a suitably experienced clinician including an HPB CNS, who has the training and authority to assess CT, or MRI results and order further investigations.
For suspected cholangiocarcinoma, clinical assessment can be undertaken by a suitably experienced HPB MDT as per British Society of Gastroenterology guidelines for diagnosis and management of cholangiocarcinoma.
Preparation for any further tests and suitability for treatment and any requirements for pre-habilitation (e.g. diabetes, lifestyle intervention, weight reviews) and pre-operative work up should be considered at this stage in the pathway and communicated to patients.
- Booking of further diagnostic testing should be carried out at the point of clinical assessment. The patient should be supported to discuss next steps and prepare for a further investigation appointment.
It is important that the healthcare professional undertaking the clinical assessment of the initial CT scan communicates with HPB specialists to determine and book next investigations. ERCP standards of care should include brushings and stent as per British Society of Gastroenterology’s ERCP – The Way Forward, A Standards Framework.
- Patients should be informed about cancer being diagnosed at the earliest face-to-face opportunity unless the patient has expressed an alternative method of communication to speed up communication. In this timed pathway, this can be done at straight to test clinic (by day 8), follow-up testing or a results outpatient appointment (by day 16), or at a treatment planning outpatient clinic (by day 21).
When cancer is diagnosed and a discussion with the patient has been held, they will be added to a waiting list for surgery, if appropriate, or seen at a cancer centre for consent and pre-operative assessment on the same day to avoid unnecessary travel and improve patient experience.
Given the speed of progression for pancreatic cancer and the rapid deterioration in physical, nutritional, and psychological health in the weeks following a diagnosis, it is important that the treatment start date is scheduled as soon as clinically appropriate following the decision to treat. For continuity of care, it is best practice for the HPB CNS from triage and diagnostics to be available in the clinic when the discussion is held.
This outpatient appointment is also an opportunity for the HPB CNS to discuss symptom management, identify physical, psychological, nutritional, or palliative care needs as well as refer for any pre-habilitation, occupational, nutritional or psychological support as required.
The OPA is also an opportunity to obtain consent from the patient to be involved in nationwide data collection initiatives that aim to build an imaging, staging and clinical data repository for future pancreatic cancer research.
When cancer is ruled out at the initially suspected site but there remains a high probability of a cancer at a different tumour site or serious non-malignant pathology (e.g. decompensated liver disease, acute liver injury), best practice would be to refer the patient to a suitable alternative department within the same provider, rather than discharging the patient back to primary care.
For example, in a patient with weight loss and abdominal pain where Pancreatic, Liver, or Gall Bladder cancer has been ruled out by a CT but another cancer is suspected, it would be appropriate for secondary care to refer directly onto an alternative site-specific pathway (e.g. the colorectal pathway), after all upper GI investigations, including endoscopy, are complete.
Alternatively in this scenario, if available, a referral to a non-site specific (NSS) pathway should be considered prior to discharging the patient back to their GP.
Cancer waiting time rules (including ‘clock start’ and ‘clock stop’) are set out in the National Cancer Waiting Times Monitoring Dataset Guidance v12.0.
- The local MDT should be used for patients where there is still uncertainty or unconfirmed suspicion. The core roles at the specialist MDT (sMDT) are lead clinician, radiologist, and pathologist, to review investigation results with a HPB or upper GI CNS and pathway navigator. The capacity required to deliver these core roles should be reflected in job plans. National guidance on how to maximise effectiveness of MDT meetings is available.
Locally agreed, clear criteria for referral to sMDT can also support with efficient pathway management. A multidisciplinary clinic with HPB expertise, including a surgeon, an oncologist, pre-operative staff, including an anaesthetist and AHPs such as a therapeutic radiographer and a specialist dietitian should be considered. This aims to improve patient experience, improve communication, and prevent delays in starting treatment.
- PET-CT service specifications set out the standards of care expected from organisations funded by NHS England to provide specialised care, including the expected timings for the provider to “appoint an examination date, perform the examination, complete the diagnostic report and return the diagnostic report and images to the referring clinician within seven business days or as specified by the contract”.
PET-CT should be carried out and reported within 10 calendar days, and by day 18, to allow preparation for a pre-arranged outpatient clinic and treatment planning discussion by day 21. NICE’s NG85 guidelines should be followed where appropriate.
- Biopsy procedures should be carried out within five days of request, and by a suitably experienced clinician, ensuring that minimum locally agreed standards are applied to allow for advanced biomarker testing / molecular profiling as outlined in the national genomic test directory and the British Society of Gastroenterology guidelines for the diagnosis and management of cholangiocarcinoma
Histopathology reports for biopsies should aim to be available in five working days and should aim to be reported ahead of the treatment planning discussion at day 21.
While this may be longer if ancillary tests are required to establish a diagnosis or if the pathway for a biopsy reaching the reporting laboratory is delayed, this should not prevent patients from moving through the pathway and receiving supportive care interventions or treatments.
- Outpatient clinics should include the HPB CNS assigned to the patient earlier in the pathway, from clinical triage (day 0 to 3) or when a likely cancer diagnosis was discussed (day 1 to 24).
Personalised care and support planning should be based on the patient and clinician(s) completing a holistic needs assessment (HNA), usually soon after diagnosis. The HNA ensures conversations focus on what matters to the patient, considering wider health, wellbeing, practical issues, and support in addition to clinical needs and fitness. This enables shared decision-making regarding treatment and care options. All patients should have access to, and information about, clinical trials. Further information about clinical trials for cholangiocarcinoma are available on the AMMF – The Cholangiocarcinoma Charity website.
Treatment decisions and plans should also be communicated back to the GP. Early consideration of the patient’s physical, nutritional, and psychological fitness for radical therapy should be addressed as soon as possible in the pathway to minimise delays in expediting treatment.
There should also be access to a HPB specialist dietitian either as part of the outpatient appointment, or if not appropriate at this clinic, arrange for a specialist dietitian to follow up with a patient to arrange a face-to-face appointment.
Local protocols and initiatives should be developed in collaboration with perioperative medicine, elderly care, and specialist dietitians. Anaesthetic assessments for patients with comorbidities should also be undertaken.
The outpatient clinic should be an opportunity early in the pathway for the HPB CNS to discuss symptom management, commence PERT, identify psychological, physical, and nutritional needs, discuss treatment plans, and facilitate pre-habilitation and pre-operative work up.
It is also an opportunity for supportive care needs assessment to be completed, recorded and referrals to allied health services (i.e. psychosocial support, occupational therapy, and pre-habilitation) actioned as required. The patient should also be referred to support services, such as psychosocial support and third sector organisations.
- Patients should be informed about other services that may support lifestyle changes, psychosocial support services, peer support networks and any voluntary services that will best address patients’ supportive care needs, such as a Specialist Pancreatic Cancer Nurse Support Line, patient information and online patient support sessions.
14. AMMF – The Cholangiocarcinoma Charity has online healthcare professional and public information at Patient Resources – AMMF.
The British Liver Trust has patient and public information
Pancreatic Cancer UK have the following online healthcare professional information and training courses to improve awareness and expertise about pancreatic cancer:
- Introduction to pancreatic cancer part one (CPD Certification)
- Introduction to pancreatic cancer part two (CPD Certification)
- Introduction to pancreatic cancer part three (CPD Certification)
- A health professional’s guide to pancreatic cancer and pancreatic enzyme replacement therapy (PERT), endorsed by the British Dietetic Association and Pancreatic Society of Great Britain and Ireland.
Pancreatic Cancer UK healthcare professional information and events
Additional information
Audit tool
This tool can be used to undertake a baseline audit of services being delivered and whether sufficient capacity is in place to routinely deliver, identify areas for improvement, select measurements for improvement, and conduct re-audits as part of continuous improvement. Patient experience measures should also be considered as part of auditing and improvement.
Table 1: Jaundice, pancreatic, extrahepatic cholangiocarcinoma and gall bladder
|
Day |
Pathway step |
Service in place? |
Capacity in place? |
|
0 |
GP referral and locally agreed minimum dataset |
|
|
|
Primary care patient information resources provided, co-developed with patients |
| ||
|
5 |
Straight to CT and bloods |
|
|
|
Commence PERT if required |
|
| |
|
Secondary care patient information resources and support provided, co-developed with patients |
|
| |
|
8 |
Clinical assessment of CT results, using PACT-UK radiology reporting pro-forma if required |
|
|
|
Booking of MRI, MRCP, PET-CT, laparoscopy / laparoscopic US as required |
|
| |
|
10 |
MDT for review and planning of diagnostic tests and potential treatment options |
|
|
|
16 |
MRI or MRCP carried out |
|
|
|
PET-CT or laparoscopy / laparoscopic ultrasound |
| ||
|
ERCP or EUS guided FNA Biopsy |
|
| |
|
Cancer ruled out or diagnosis confirmed with patient, further support and signposting provided |
|
| |
|
21 |
Treatment and care options discussed at multi-disciplinary outpatient clinic |
|
|
Table 2: Hepatocellular carcinoma and intrahepatic cholangiocarcinoma
Day | Pathway step | Service in place? | Capacity in place? |
|---|---|---|---|
|
0 | GP referral and locally agreed minimum dataset |
|
|
|
Primary Care patient information resources provided, co-developed with patients |
| ||
|
5 |
Straight to Ultrasound and bloods |
|
|
|
Multiphase CT or Contrast MRI |
|
| |
|
Secondary care patient information resources and support provided, co-developed with patients |
|
| |
|
8 |
Clinical assessment of CT or MRI results |
|
|
|
Booking of Multiphase CT, PET-CT or Contrast MRI as required |
|
| |
|
10 |
MDT for review and planning of diagnostic tests and potential treatment options |
|
|
|
16 |
Contrast MRI carried out (if not already carried out by day 3) |
|
|
|
Multiphase CT (if not already carried out by day 3), or PET-CT carried out |
| ||
|
Persutaneous Biopsy, EUS guided FNA Biopsy, and/or ERCP and Cholangioscopy |
|
| |
|
Cancer ruled out or diagnosis confirmed with patient, further support and signposting provided |
|
| |
|
21 |
Treatment and care options discussed at multi-disciplinary outpatient clinic |
|
|
Cancer Alliance workspace
Cancer Alliances access this workspace for national guidance, resources, and to share learning. Please use this space to upload materials you have developed locally and that you think would be useful for colleagues implementing this pathway across the country.
Acknowledgements
This guidance was developed by the NHS Cancer Programme and builds on experience and expertise provided by the HPB Task and Finish Group membership, including:
- Clinical representatives: Paula Ghaneh, Alison Broughton, Stephen Ryder, Giles Toogood, Jaypal Ramesh, Ed Britton, Vicki Stevenson-Hornby, Rachel Brown, Raneem Albazaz, Keith Roberts, Ganesh Radhakrishna, Dan Palmer, Tina George, Vijay Patel, Aileen Marshall
- operational representatives: Michael Ryan, Claire Goldrick
- patient and charity representatives: Anna Jewell, Peter de Rosa, Helen Morement, Pamela Healy, Charles Czajkowski
- NHS Cancer Programme